- Author: Shimat Villanassery Joseph
Save the Date!
Entomology Seminar 2015:
Tuesday, December 1, 2015
8:00 AM to 12:00 PM
County of Monterey Agricultural Center
Conference Room
1432 Abbott Street, Salinas, California
This seminar will focus on a broad range of topics dealing with arthropod pest management in coastal California, and current issues affecting growers, pest control advisors, and other agricultural professionals.
Registration/sign-in is from 7:30 to 8:00 AM. There is no fee for this meeting. Continuing education credits will be requested. Please call ahead (at least 24 hours) for arrangements for special needs; every effort will be made to accommodate full participation. For more information, contact Shimat Joseph (831-229-8985; 1432 Abbott Street, Salinas, California 93901).
Requirement from California DPR: Bring your license or certificate card to the meeting for verification when signing in for continuing education units.
- Author: Shimat Villanassery Joseph
Cabbage maggot (Delia radicum) is a serious insect pest of Brassica crops such as broccoli and cauliflower in the Central Coast of California. These crops are grown throughout the year; as a result cabbage maggot problems persist year long.Cabbage maggot eggs are primarily laid in the soil around the crown area of the plant. A single female fly can lay 300 eggs under laboratory conditions. The eggs hatch within 2-3 days and the maggots feed on the taproot for up to three weeks and can destroy the root system of the plant. The maggots pupate in the soil surrounding the root system and emerge into flies within 2-4 weeks. Severe cabbage maggot feeding injury to the roots cause yellowing, stunting even plant death.
Control of cabbage maggot on Brassica crops primarily involves the use of soil applied organophosphate insecticides such as chlorpyrifos and diazinon. However, the persistent use of organophosphate insecticides has resulted in high concentrations of the insecticide residues in the water bodies posing risks to non-target organisms and public health through contaminated water. Currently, use of organophosphate insecticides is strictly regulated by California Department of Pesticide Regulation. There is therefore an urgent need to determine the efficacy of alternate insecticides for cabbage maggot control.
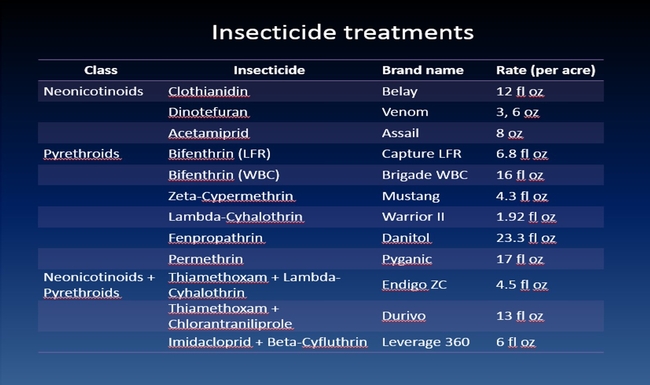
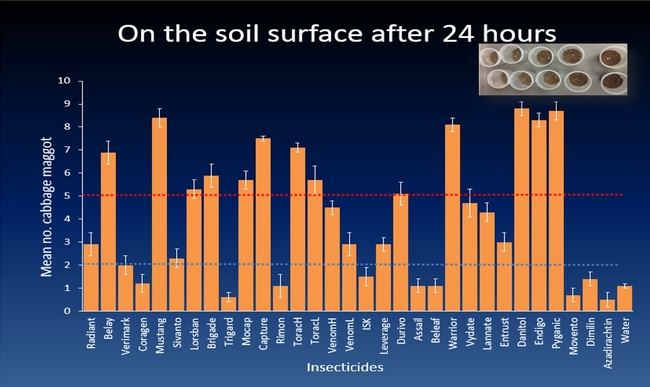
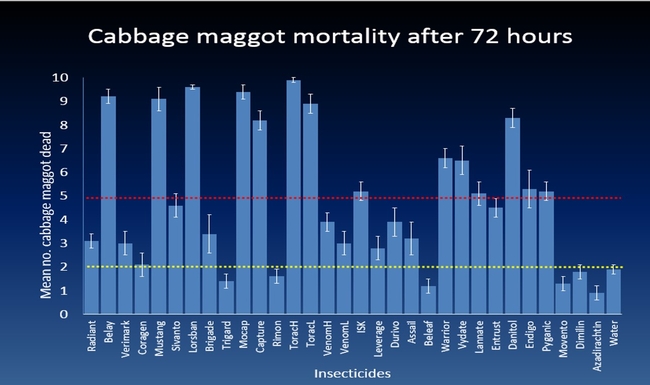
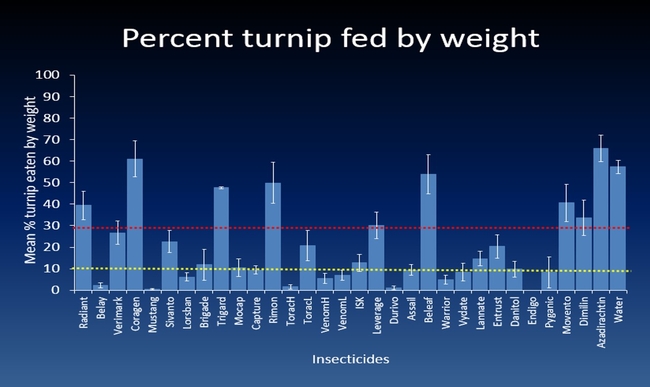
The efficacy of 29 insecticides was determined against cabbage maggot through a laboratory bioassay by exposing field collected maggots to insecticide treated soil immediately after application. Three parameters were used to evaluate efficacy (1) proportion of maggots on the soil surface after 24 h, (2) proportion of change in weight of turnip bait, and (3) dead maggots after 72 h. Based on the assays, 11 insecticides performed better and they were Mustang, Torac, Danitol, Belay, Capture, Warrior II, Lorsban, Mocap, Durivo, Pyganic and Vydate in the order of highest to lowest efficacy. Eight insecticides were selected based on superior efficacy to determine the length of residual activity on cabbage maggot larvae. The persistence of insecticide activity was greater with Capture, Torac and Belay than with other insecticides tested.
The mode of exposure of insecticides in this study was entirely by contact (through skin) and other modes of exposure such as ingestion (through mouth) or through respiratory holes (spiracles) were not investigated. Some of the insecticides tested in the study were insect growth regulators (IGRs) (Dimilin, Rimon, Trigard, and Aza-direct), which normally interfere with the growth and development of the insect and they showed a low efficacy against cabbage maggot larvae. Entrust (spinosad) showed a moderate efficacy possibly because the primary mode of exposure to Entrust is by ingestion. The diamide insecticides (Beleaf, Coragen and Verimark) have systemic activity as they move within the plant and likely away from the site of application. It is possible that the soil applied diamide insecticides are absorbed by the roots and translocated to the above ground plant parts with little effect on the feeding larvae in the tap roots.
This study was conducted under controlled conditions in the laboratory and the results may not be entirely consistent in field conditions. The Brassica fields in the California's Central Coast are profusely sprinkler irrigated up to three weeks after sowing to ensure uniform germination and proper establishment of plants. It is likely that applied insecticides are partially or completely leached out of the root zone area without providing anticipated maggot control. In this study, insecticides were drenched into the cup and none of the applied insecticide solution leached out. Therefore, it is likely that the insecticides were more effective in the laboratory assay than they would be in the field. Certain insecticides such as pyrethroids tend to bind to the soil organic matter. The organic matter in the California's Central Coast soils can be up to 4%, which could reduce the availability of soil applied pyrethroid insecticide to the root zone where cabbage maggot larvae typically colonize. In situations with poor insecticide spray coverage, invading cabbage maggot larvae are possibly exposed to no or sub-lethal doses of the soil applied insecticide and may be able to penetrate the soil and infest the roots. The air temperature in the field at the time of insecticide application may influence the efficacy of the applied insecticide. The efficacy of pyganic decreased as the temperature increased against onion maggot. This suggests that application of pyrethroid insecticides should be avoided during warmer periods of day.
Other field conditions that influence efficacy of insecticides are cabbage maggot incidence and frequency of invading cabbage maggot flies on Brassica crop in the Central Coast of California. The earliest peak of cabbage maggot infestation occur a month after sowing broccoli seeds and infestations can be continuous until harvest. Also, insecticides applied at sowing as a banded spray on the seed lines did not provide adequate cabbage maggot control based on the insecticide efficacy trials conducted in commercial broccoli fields. These findings suggest that delaying the insecticide application by 2-3 weeks after sowing is more likely to maximize maggot control. Because the cabbage maggot infestation can last several weeks, insecticides with extended persistence of efficacy would increase the value for cabbage maggot control. Overall, results show that Capture, Torac and Belay which performed effectively against cabbage maggot for a month after application. This indicates that insecticides used before the first peak of infestation may protect the younger stages of the Brassica plants allowing them to establish and tolerate milder cabbage maggot infestations thereafter.
In conclusion, 11 insecticides with high efficacy were identified for future investigation. Future studies will focus on determining the effects of application timing and delivery methods compatible with cabbage maggot incidence in both directly sown and transplanted Brassica crops in the Central Coast of California.
If you are interested in reading the details of this study, please click the link below to access the published article.





- Author: Shimat Villanassery Joseph
- Author: Mark Bolda
Lygus bug (Lygus herperus) (Figure 1a) continues to be a major pest of strawberry in northern Central Coast of California. The feeding injury on the young developing fruits results in catfaced fruits (Figure 1b) rendering them unmarketable. Management of lygus bug in strawberry is always been a challenge because of lack of effective insecticides with desirable attributes such as short pre-harvest intervals (PHIs). Moreover, the insecticides registered on strawberry for lygus bug control have been used over and over in the same season and it is likely that lygus bug developed resistance to those insecticides.
In 2015, an insecticide trial was conducted to evaluate the efficacy of newer insecticides registered on lygus bug. The details on insecticide products, active ingredients and rates are shown in Table 1. The newer insecticides tested were Sivanto and Sequoia. Sivanto is registered on strawberry with the maximum rate 14 fl oz per acre. Sequoia is not registered at this moment and rate tested is 2.88 fl oz per acre which is lower than 4.5 fl oz or 5.75 fl oz per acre tested in the previous years. The study was a replicated (5 replications) and the treatments were randomized. The plot size was ten 65-feet long beds which is fairly large for insecticide trial in a commercial strawberry field. First broadcast-spray application of insecticides was done on 13 June 2015 followed by a second broadcast-spray application on 20 June 2015. The insecticides were applied using commercial tractor mounted sprayer. The water volume used for both the applications was 200 gal/ acre. Dynamic (surfactant) was added at 0.25% v/v.
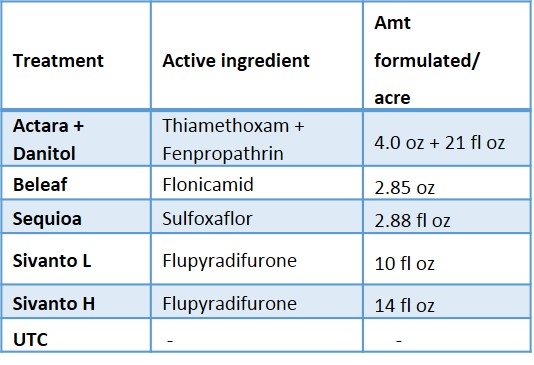
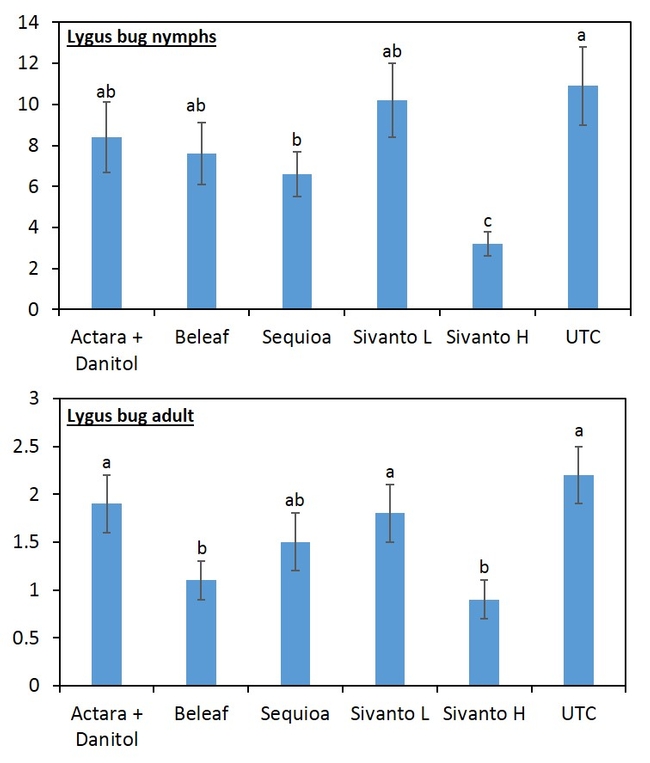
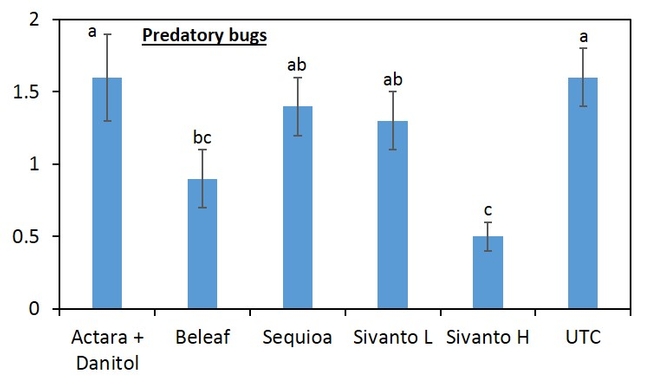
Beat-trays were used to sample insect populations (Figure 2). Twenty strawberry plants were sampled and the sampling consists of five strikes per plant with the lid of a regular sized Rubbermaid container. Sampling was done a day before application then at 3 and 7 days after first application then 3, 7, 14, 21, and 28 days after second application. The insect samples were bagged, transported to the laboratory and stored in the freezer for later evaluation in the laboratory. The samples were evaluated for all nymph stages and adult of lygus bug, thrips, predators (damsel bug, minute pirate bug, bigeyed bug, rove beetle, and spiders) (Figure 3) and parasitoids. In addition, 100 fruits were randomly sampled from each plot at 28-days after second insecticide application. The fruits were evaluated for lygus bug injury or “catface” and other unmarketable symptoms such as rot, spit strawberries etc.
When all the data were combined, number of lygus bug nymphs were lower in the higher rate of Sivanto and Sequoia than in untreated check treatment (Figure 4). Lower number of lygus bug adult was captured in higher rate of Sivanto and Beleaf than in untreated check. Similarly, number of predatory bugs was lower in the higher rate of Sivanto than in other treatments. Spiders captured were similar among treatments (Figure 5).
On fruit evaluation, there was no difference in number of fruits with catface injury or those marketable fruits among the insecticide treatments, although numerically, number of fruit with catface injury was lower in the higher rate of Sivanto treatment than in other treatments.
Overall, it appears that Sivanto at 14 fl oz per acre performed better than other treatments against lygus bug. Sequoia and Beleaf also showed evidence of lygus bug suppression. However, Sivanto at 10 fl oz per acre did not suppress lygus bug. The representative industry standard -- combined treatment of Actara and Danitol did not show any evidence of lygus bug suppression in this study.
If you are interested in reading the complete report, please click the link below.



- Author: Larry J Bettiga
Larry Bettiga, Viticulture Farm Advisor, UC Cooperative Extension, Monterey, San Benito and Santa Cruz Counties
Grapevine leafroll and red blotch disease are two virus-associated diseases that should be on the radar of all grape growers. The following article will hopefully provide you an update on these virus diseases base on our current knowledge. Surveying vineyards during harvest is a great time to assess vineyard blocks for the presence of disease symptoms.
Grapevine Leafroll Disease
Leafroll is one of the more important virus diseases of grapevines. It occurs in every major grape growing area of the world. There are five grapevine leafroll associated viruses (GLRaVs) that are serologically distinct. These single stranded RNA viruses are placed in a family called Closteroviridae. The majority of these are grouped in the genus Ampelovirus (GLRaV-1, -3, and -4), most of the viruses in this genus have been demonstrated to be vectored by mealybugs and scale insects in vineyards. GLRav-2 is in the genus Closterovirus, and GLRaV-7 is in the genus Velarivirus, there is no known vector of these two genera.
These viruses can cause similar symptoms in infected grapevines. All the GLRaVs can be transmitted by vegetative propagation and grafting; GLRaVs in Ampelovirus can also be transmitted by the mealybugs and soft-scale insects in vineyards. GLRaV-3 is the predominant species found in most vineyards worldwide. Recent surveys in the north coast have shown 80% of symptomatic vines sampled were infected with GLRaV-3.
To further complicate matters there are variants that have been identified for given GLRaV species. For GLRaV-3 there are several distinct variants known to exist. What needs to be better understood is the significance of these GLRaV-3 variants and their interactions with other viruses when multiple infections exist in a vine. For GLRaV-2 the “Red Globe” variant is known to cause graft incompatibility when grafted onto certain rootstocks (5BB, 5C, 3309C and 1103P) resulting in the decline and death of vines.
In the post-phylloxera infestation plantings that have occurred on the central coast during the past 20 years there has been an increased incidence of grapevine leafroll disease. The use of non-certified scion material has been a major contributor to this disease increase. The other issue has been the spread of leafroll (primarily GLRaV-3) from infected vineyards to adjacent vineyards planted with California-certified stock. UC research documented the rapid spread of leafroll into a certified planting from an adjacent infected block. During the 5 years of observation the annual rate of increase in leafroll symptomatic vines was more than 10% in a Napa Valley site.
Recognizing Leafroll
Leaf symptoms become visually apparent by early summer and generally intensify into midsummer and fall. Physical stresses to the vine may increase symptom severity and there are similar symptoms caused by other abiotic and biotic injuries. On affected vines, the margins of the leaf blades roll downward, starting with the basal leaf on the cane. Areas between the major veins turn yellow or red, depending on whether the cultivar produces white or red fruit. In some cultivars, the area adjacent to the major veins remains green until late fall.
The most important effect of leafroll disease is a reduction in the yield and quality of fruit from infected vines. Yield losses of 10 to 20% are fairly typical. Because leafroll viruses damage the phloem of infected vines, sugar accumulation is delayed and color pigment production is reduced. Fruit from infected vines can be low in sugar, poorly colored, and late in ripening.
It is important to remember that the lack of symptoms in a grapevine does not guarantee freedom from infection by the viruses that are the causal agents of leafroll disease.
Leafroll disease on Pinot noir (top) showing burgundy red between green main leaf veins accompanied by downward rolling of the leaf margins; on Chardonnay (bottom) leaves show a more generalized chlorosis and downward rolling of the leaf margins in late fall.
Lab Testing
Leafroll viruses may be diagnosed using ELISA and RT-PCR tests. Virus titer levels are variable not only within the year, but also within the vine. Collect petioles in late summer and fall, or shoots/canes for cambium scrapings in fall and winter. PCR and ELISA tests are not available for all GLRaVs. Check with the commercial lab for their preferred sampling method and collection time prior to taking samples.
Mealybug Vectors
The most common mealybug found in California vineyards is the grape mealybug (Pseudococcus maritimus). Obscure mealybug (P. viburni) is present in central coast vineyards but less common than the grape mealybug. The vine mealybug (Planococcus ficus) was introduced into California in 1994 and has now been found in most production area of the state. Less common is the long-tailed mealybug (P. longispinus) found primarily in the cooler areas of the south central coast. The Gill's mealybug (Ferrisia gilli) is the fifth species found in California but is currently very limited in distribution with populations found in the Sierra foothills, in the northern coast (Lake County) and in the southern San Joaquin Valley.
All the above species are capable of being a vector for leafroll disease. Research has shown that mealybugs can become infective after one hour of feeding on a leafroll virus infected vine and can transmit the virus to a clean host after one hour of feeding. Although all female instars can transmit the virus once infected, the first instar is the most effective vectors of the disease. The first instar or “crawler” moves to find a feeding spot and is considered to be the most common dispersal stage of a mealybug population. Wind, equipment, workers and infested nursery stock can also move mealybugs.
Movement of leafroll disease into a recently planted vineyard of certified planting stock from an infected block on the opposite side of the avenue. Note the vines showing symptoms are nearest the avenue and are not present on vines further down the rows.
Management of Grapevine Leafroll Disease
1. Plant Material.The first management strategy should be to plant certified vines that have been grown and produced by a nursery participating in the California Grapevine Registration and Certification Program. Once virus infected a vine will remain infected, there is no cure. Commercial nurseries that produce certified grapevines and participate in the California Grapevine R&C Program obtain their clean stock from the Foundation Plant Services at the University of California, Davis. UC Davis has a foundation vineyard for major grape cultivars and clones. Before being planted in the foundation vineyard, all vines are tested across biological indicators, and by ELISA and RT-PCR. The foundation vineyard is monitored by visual inspections in spring and fall, and a portion of it is retested every year by ELISA and RT-PCR for viruses known to spread naturally. This provides the highest level of confidence about the virus status of the selections.
Both the fruiting scion and the rootstock need to come from certified mother plants. A very common spread of leafroll is the use of infected bud wood from commercial vineyards. The lack of symptoms in the source vineyard cannot be relied upon as a guarantee that there is no virus; many of the major grapevine viruses show no symptoms during some or all of the season. Particularly if wood is collected during the dormant season, it is unlikely that the source vines will show distinct symptoms of virus infection. Selected grapevines should also be pre-tested for virus by a competent diagnostic laboratory if this type of material is going to be used. Even with vine testing sourcing bud wood from established vineyards carries a risk of introducing virus into a new planting.
2. Learn to recognize leafroll symptoms.Leafrollsymptomsbecome visually apparent by early summer and generally intensify into midsummer and fall as noted above. Symptoms can vary by leafroll species, multiple virus infections, and by cultivar and rootstock combination. Symptoms are generally more apparent in cultivars producing red or black fruit than in white fruiting cultivars. Remember that the lack of symptoms in a grapevine does not guarantee freedom from infection by the viruses that are the causal agents of leafroll disease.
3. Recognize and be aware of potential leafroll vectors.As discussed above mealybugs and scale insects are known vectors of some species of GLRaVs. Monitor and be aware of which insect vectors may be in your vineyards. More information on these insects is available in Grape Pest Management UCANR publication 3343 or in the online UC IPM guideline for grapes, http://www.ipm.ucdavis.edu. Know which species of mealybugs are present in your vineyards, their population dynamics are different and will influence the timing of any needed control practices. European fruit lecanium scale (Parthenolecanium corni) is a common insect found in California vineyards, it and other scale insects has been shown to transmit some GLRaV species.
4. Be aware of potential spread from leafroll infected blocks.Leafroll infected blocks can be a source for vector and disease spread into adjacent clean plantings. Consider if plant removal is a viable option to reduce further spread for both the source and clean blocks. Vector control may be a management decision to consider. Recent research suggests the rate of disease spread of GLRaV-3 is greater when higher mealybug population levels are present. Treatment of virus source blocks should minimize the infective vectors leaving the block; the treatment of clean blocks should be targeted to kill infective vectors quickly upon entering the block and to reduce secondary spread to adjacent vines.
5. Area-wide management.When both mealybug populations and the virus causing leafroll disease are present in an area, cooperation between neighboring vineyard owners will be necessary to improve on reducing the spread of disease from infected source blocks to non-infected vineyards.
Grapevine leafroll disease is actively being studied both here in the US and internationally. Improvements in identification techniques and better understanding of disease epidemiology in vineyards will hopefully improve our ability to develop management practices to reduce economic impacts.
Red Blotch Disease
Grapevine red blotch disease was suspected as a concern on vines growing in the Napa Valley in 2008. In 2011 a DNA virus was identified in independent studies in California and New York and shown to be associated with the symptoms on infected vines. Since the initial identification of Grapevine red blotch-associated virus (GRBaV), it has been found to be widespread in vineyard producing areas of the United States and Canada. A recent survey of a grape herbarium collection at UC Davis has shown one plant specimen collected by Harold Olmo in 1940 from a Sonoma County vineyard to be positive for GRBaV.
Recognizing Red Blotch
In red wine cultivars, irregular red blotches form on the leaf blades on the basal parts of shoots. Veins on affected leaves can turn pink to red in color. Symptoms can vary between cultivars and the severity may also vary between years. In white cultivars the symptoms are not as dramatic. Interveinal chlorosis is most common followed by irregular chlorotic blotches. These symptoms can begin to appear as early as July and as late as September. In comparing red blotch to leafroll disease, leafroll symptoms are generally more uniform across the leaf blade, the veins remain green, and the there can be a downward rolling of the leaf margin. For more pictures of red blotch symptoms on different cultivars got to: GRBaV symptoms
Late summer symptoms on Chardonnay, red blotch on the upper photo and leafroll on the bottom.
Lab Testing
The identification of GRBaV can be difficult to determine by visual observation due to the similarity of the symptoms to leafroll disease and other nutrient deficiencies. This is especially true in the case of white cultivars. Co-infections with other viruses can also affect symptom expression. Suspected infections should be confirmed by having samples assayed by a PCR test by a commercial diagnostic lab. Check with the commercial lab for their preferred sampling method and collection time prior to taking samples.
Disease Spread
GRBaV is spread by the propagation of planting stockor grafting non-infected vines using infected budwood. The widespread occurrence of red blotch disease would suggest this type of spread has occurred. Since the identification of the virus and the availability of the PCR testing in 2012, grapevine nurseries have been testing their increase blocks and removing infected vines to eliminate this type of spread. The recently established Russell Ranch Foundation Vineyard at UC Davis has been tested and all vines are free from GRBaV. Each vine planted at the Russell Ranch has undergone extensive virus testing following a process known as Protocol 2010. For nurseries participating in the CDFA R&C Program this will provide a source for future increase blocks to supply certified vines for vineyard plantings.
GRBaV is a DNA virus and is closely related to a family of viruses called Geminiviridae. Insect such as leafhoppers and whiteflies can vector other virus diseases within this family. Researchers are currently testing potential insect vectors of GRBaV. Although there was a report of Virginia creeper leafhopper being a vector in a greenhouse study other researchers have been unable to duplicate that study. The evaluations of other potential insect vectors have not yet identified one that can successfully acquire and transmit the virus in the field. Although there is anecdotal information that there is spread within some vineyards to date there is insufficient data to support that claim.
Vine Effects
Research has shown that when comparing GRBaV infected vines to ones that have no known GRBaV, leafroll-associated viruses, vitiviruses, or Nepoviruses that Brix were lower and malic acid in the juice were higher at harvest for Cabernet Sauvignon and Chardonnay but not Zinfandel. For Chardonnay, yield was also reduced for infected vines. A study looking at the effect of dropping crop to improve quality on infected vines saw little beneficial effect from that practice. For most cultivars, there is a decrease in total phenols, tannins, and anthocyanins (for red wine cultivars) for vines infected with GRBaV.
Management of Red Blotch Disease
As with leafroll disease the first management strategy should be to use propagation material that is free from known viruses when establishing new vineyards or grafting existing sites. Meetings of the Grapevine Regulations Working Group have been recently conducted to discuss proposed changes to the Grapevine Registration & Certification Program with regards to red blotch disease. Until budwood increased from the Russell Ranch Foundation vineyard is available for use it is important that propagation material is tested to avoid virus disease.
If you have blocks that have leaf symptoms and have had delayed maturity or low crop yield have a virus panel run by a commercial lab to confirm which viruses are present. Remember symptoms are going to be more noticeable in red wine cultivars and less so with white cultivars. For confirmed GRBaV infected vineyards the management response may vary depending on the vine effects that are being observed. The difference in sugar accumulation between infected and non-infected vines in some vineyards has been as much as 5 Brix. In vineyards with a combination of infected and non-infected vines this wide variation in maturity has resulted in selective harvests to improve fruit uniformity. For infected sites that fail to meet yield and quality expectations vineyard removal is the best solution. If only a low percent of vines in a block are infected, then rogueing and replanting is an option. If vine removal and replanting is an option there is currently assistance available (see below).
Financial Assistance Available to Replant Red blotch-affected Grapevines:
The Agricultural Act of 2014 (the 2014 Farm Bill) authorized the Tree Assistance Program (TAP) to provide financial assistance to qualifying orchardists and nursery tree growers to replant or rehabilitate eligible trees, bushes and vines damaged by natural disasters.
The 2014 Farm Bill makes TAP a permanent disaster program and provides retroactive authority to cover eligible losses back to Oct. 1, 2011. In California, producers who are replanting vines affected by grapevine red blotch disease may be eligible for assistance and should contact their local Farm Service Agency Office to schedule an appointment for the required visual inspection. For more information go to: Fact Sheet
References
Al Rwahnih, M., et al. 2013. Association of a DNA virus with Grapevines affected by Red Blotch disease in California. Phytopathology 103:1069-1076.
Daane, K. M. et al. 2012. Biology and management of mealybugs in vineyards. p. 271-307. In: N. J. Bostanian et al. (eds.), Arthropod Management in Vineyards: Pests, Approaches and Future Directions. Springer. 505p
Golino, D. A., et al. 2002. Grapevine leafroll disease can be spread by California mealybugs. California Agriculture 56:196-201.
Golino, D. A., et al. 1992. Grapevine virus diseases. In: Bettiga, L, (ed.), Grape Pest Management, 3rd ed. Oakland: University of California Division of Agriculture and Natural Resources, Publication 3343, 157-173.
Golino, D. A., et al. 2008. Leafroll disease is spreading rapidly in a Napa Valley vineyard. Calif. Agric. 62:156-160.
Krenz, B., Thompson, J., et al. 2012. Complete Genome Sequence of a New Circular DNA Virus from Grapevine. J. Virol. 86:7715.
Krenz, B., et al. 2014. Grapevine red blotch-associated virus is widespread in the United States. Phytopathology. First Look.
Oberholster, A., et al. 2015. Impact of red blotch disease on grape and wine composition and quality. American Society of Enology and Viticulture National Conference Technical Abstracts, p. 75. (2015).
Poojari, S., et al. 2013. A leafhopper transmissible DNA virus with novel evolutionary lineage in the family Geminiviridae implicated in grapevine redleaf disease by nextgeneration sequencing. Plos One 8:e64194.
Sharma, A. M. et al. 2011. Occurrence of grapevine leafroll-associated virus complex in Napa Valley. PLoS One 6(10): e26227.
Smith, R., et al. 2015. Effect of crop reduction of vines infected with grapevine red blotch-associated virus on fruit maturity. American Society of Enology and Viticulture National Conference Technical Abstracts, p. 136-137.
Sudarshana, M. and M. Fuchs. 2015. Grapevine red blotch In: Wilcox, W., et al, (eds.), Compendium of Grape Diseases, Disorders and Pests, 2nd ed. The American Phytopathological Society.122-123.
Tsai, C. W., et al. 2010. Mealybug transmission of grapevine leafroll viruses: Analysis of virus–vector specificity. Phytopathology 100:830-834.
Authors: Richard Smith1, Michael Cahn1, Tamara Voss2, Toby O'Geen3, Eric Brennan4, Karen Lowell5 and Mark Bolda6
1 – UC Cooperative Extension, Monterey County; 2 – Monterey County Water Resources Agency; 3 – Dept of Land Air and Water Resources, UC Davis; 4 – USDA Agricultural Research Service; 5 – Natural Resources Conservation Service; 6 – UC Cooperative Extension, Santa Cruz County.
For access to full report please vist http://cemonterey.ucanr.edu/files/219694.pdf
Summary: After four years of drought, groundwater levels in the Salinas Valley are at historically low levels which threaten to adversely affect farming in the Salinas Valley. Given the prospect of a strong El Niño this coming winter, it seems prudent to plan to capture as much of the rainfall as possible to maximize infiltration into the soil and recharge groundwater. The east side of the Salinas Valley is particularly hard hit because it receives a lower proportion of recharge from the Salinas River than other hydrological subunits in the Salinas Valley and its water levels have fallen to lower levels. Low residue cover crops have been shown to effectively increase rainwater infiltration on Chualar loam soils and they provide a practice that we can employ to increase groundwater recharge. Given the low water status of the aquifers and the forecast for significant rainfall, there is an opportunity to proactively implement practices that can maximize water capture this winter. In this article we discuss practices growers can employ in production fields and on farm edges to maximize rainwater infiltration and restore groundwater resources.
Introduction: The four years of drought have left groundwater levels in much of the Salinas Valley are at historically low levels (Figure 1). Due to the types of sediments in Salinas Valley, it is unlikely that the low groundwater levels will cause land to subside; however other deleterious effects may result. The most immediate effect is that shallower wells may become unreliable (i.e., either produce less water or suck in air or sand). This is an immediate expense for growers, who will need to service these wells or drill deeper. As groundwater levels drop below sea level, the salty ocean water will move into coastal aquifers worsening current seawater intrusion issues. Although tremendous efforts have been made in the Salinas Valley to stop seawater intrusion, the prolonged drought increases the potential for seawater to move inland. Groundwater levels on the east side of the Salinas Valley are particularly concerning because they are declining the fastest, which has set up a gradient for saline groundwater to flow toward this part of the valley.
It is encouraging that El Niño conditions continue to persist in the eastern Pacific, and may lead to significant precipitation on the Central Coast this winter. The challenge is to infiltrate as much of this rain as possible to help recharge the groundwater, rather than allow it to escape as run-off into the ocean. Much of the east side agricultural land has moderate to excellent ability to infiltrate rainfall which will directly recharge the underlying aquifer. For example, infiltrating an additional 4 inches of rainfall per acre across 100,000 acres could potentially add 33,000 acre-ft of recharge during the winter. Enhancing recharge in areas with declining groundwater levels would be especially beneficial in reducing seawater intrusion. Increasing the infiltration during severe storm events would also lower flood risk and erosion damage. The objective of this article is to briefly review potential strategies to increase infiltration during the winter storms.
East side Hydrology: Inflows to the Salinas Valley groundwater basin are estimated at 504,000 acre feet/year during an average rainfall year, with about 50 percent from stream recharge (including Nacimiento and San Antonio reservoir releases), 44 percent from deep percolation from precipitation and agricultural return flows, and 6 percent from subsurface inflow from adjacent groundwater basins (MW, 1998). Groundwater recharge in Forebay, Pressure, and Upper Valley subareas of the Salinas Valley Groundwater Basin are primarily from infiltration from the Salinas River. Releases from San Antonio and Nacimiento reservoirs flow down the river recharging the aquifer of these hydrological regions.
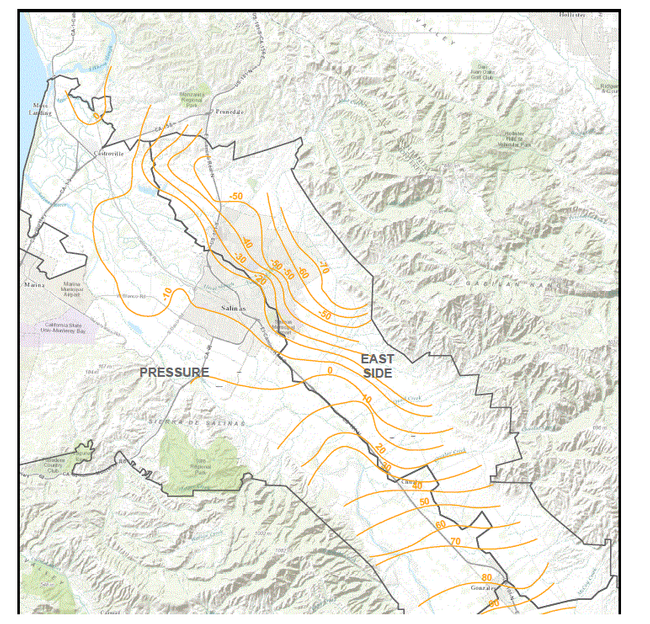
Inflow to the East Side Subarea results from a combination of infiltration along small streams on the west side of the Gabilan Range, direct recharge by precipitation on the valley floor, and subsurface inflow from the Pressure and Forebay Subareas (Brown and Caldwell, 2015). In the East Side Subarea, Shallow Aquifer, the seasonal pattern of groundwater head elevation changes are correlated most strongly to annual precipitation (Brown and Caldwell, 2015). In wells with perforations in both the east side shallow and deep aquifers, fall groundwater head generally follows the pattern of cumulative precipitation surplus, with head declining during relatively dry periods and rising during relatively wet periods. There is, however an overall long-term decline in groundwater head over the period of record (1953-2013) for the East Side subarea (Figure 1) (Brown and Caldwell, 2015). Groundwater levels on the east side of the Salinas Valley are particularly concerning because they have set up a gradient for saline groundwater to flow toward this area of the Valley (Figure 2). The cumulative storage change for the East Side subarea has also been negative for the entire period of record (1944-2013), investigated in “State of the Salinas River Groundwater Basin” report, ending at about -332,600 acre-feet in 2013. (Brown and Caldwell, 2015)
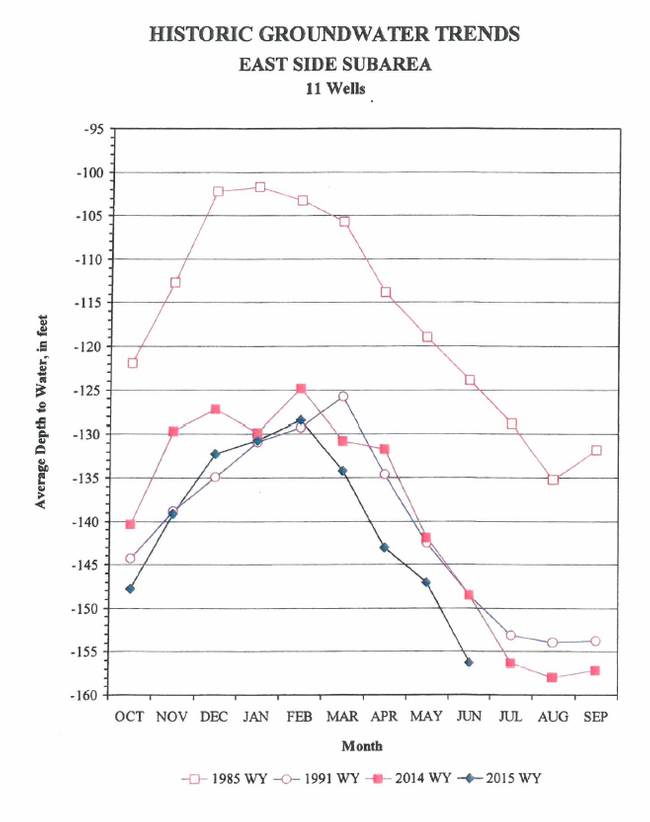
Figure 2. Groundwater head elevation in the Pressure 180-foot and East Side shallow aquifers. Source: Monterey County Water Resources Agency; Map date August 20, 2015.
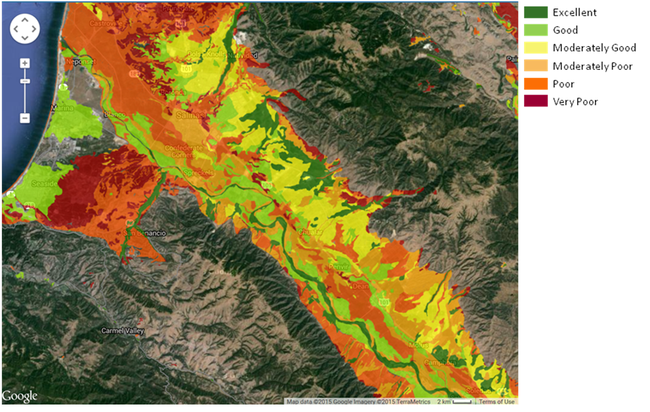
Soil Types and Groundwater Recharge Potential: Common soil series on the east side of the Salinas Valley include Chualar, Antioch, Arroyo Seco, Danville, Elder and Placentia. The Chualar series is the most common soil type and has moderately good potential for infiltrating water from winter storms (Table 1, Figure 3). However, it has a tendency to form a crust and shed water during high intensity rainfall events. Other common soils such as Arroyo Seco, and Elder are highly permeable and have excellent potential to recharge groundwater. Soils such as Antioch are poorly suited, but deep tillage increases the permeability of subsoil horizons and greatly improve its recharge capability. Soils such as Placentia, Danville and Salinas have slow percolation rates. These soils have fine texture throughout the profile yet improving soil structure could increase their ability to accept water.
Table 1. Summary of groundwater recharge potential ratings of common soils on the east side of the Salinas Valley based the Soil Agricultural Groundwater Banking Index (O'Geen et al., 2015).
|
Soil Series |
Groundwater Recharge Potential Rating |
|
|
|
No Deep Tillage |
Soil Modified by Deep Tillage |
|
Chualar |
Moderately Good |
Moderately Good |
|
Antioch |
Poor |
Good |
|
Arroyo Seco |
Excellent |
Excellent |
|
Placentia |
Poor |
Poor |
|
Danville |
Moderately poor |
Moderately poor |
|
Elder |
Excellent |
Excellent |
|
Gloria |
Very poor |
Moderately poor |
|
Pico |
Good |
Good |
|
Rincon |
Poor |
Poor |
|
Cropley |
Poor |
Poor |
|
Salinas |
Poor |
Moderately poor |
Figure 3. Suitability of east side soils (modified by deep tillage) for groundwater recharge (O'Geen et al., 2015).
Practices to Increase Rain Water Infiltration: The overall approach to increase infiltration on agricultural land during winter storms is to implement practices that maximize infield infiltration in conjunction with practices on field edges which slow and retain run-off so that it has chance to infiltrate.
IN FIELD PRACTICES
Full-Season Cover Crops: Cover crops can improve the infiltration of water into the soil by protecting the soil surface, eliminating surface crusting caused by the impact of raindrops and maintaining aggregate stability and creating soil macropores. Full-season cover crops are those that are planted in the fall and incorporated into the soil in late winter or early spring. They increase infiltration by creating root pathways that facilitates downward movement of water. In addition, they slow the movement of water over the soil surface giving it more time to infiltrate. Downward movement of rainwater through the soil leaches salts that have accumulated over the growing season and contributes to groundwater recharge. However, full-season cover crops remove water from the soil by their transpiration later in the growth cycle and, in low rainfall years, they may dry the soil and little recharge will occur. For instance in a trial conducted from 2010-11, we observed 5.5 inches of water percolated into the soil in the bare fallow treatment, but only 3.0 inches in the full-term cover crop treatment (incorporated into the soil in March). The difference in the two treatments was due to evapotranspiration by the cover crop later in February and March when day length increases. This is good for reducing nitrate leaching, but less useful for groundwater recharge. Given the current drought situation and a pressing need to facilitate groundwater recharge, understanding the impact of cover crops on infiltration and options to manage them in Salinas Valley settings is critical. Low residue cover crops offer a unique approach to address operational constraints that may make full season cover crops impractical in the vegetable cropping operations that cover much of the land in the Salinas Valley.
Low Residue Cover Crops in Vegetables: Low residue cover crops are planted in the fall and killed 60 days later (e.g. mid-January) when they have produced about 0.5 tons/acre of dry biomass. This is typically when they've produced about 10 to 20% of the potential biomass of a full-season cover crop. Typical varieties used in vegetable production systems for low residue cover crops include cereals like rye (Merced and AGS104), as well as winter-dormant triticales (Trios 888). Both are typically sown in the fall following listing; earlier planting dates can provide protection to the soil for early storms in November and December.
Seeding can be done by dribbling the seed onto the furrow bottom, followed by shallow harrowing. It is important to use sufficient seed (e.g. 80-100 lbs rye seed/acre) to get rapid growth that can quickly protect the soil from early storms. Planting seed in the furrow is tricky because soil can fall from the edge of the bed back into the furrow (after the harrow passes) and bury the seed too deeply. The ideal seeding depth is 1-2 inches; four inches is too deep and will greatly reduce seed emergence. Under ideal conditions the soil may be moist enough to germinate the seeds without irrigation, or an early rain provides the moisture for germination.
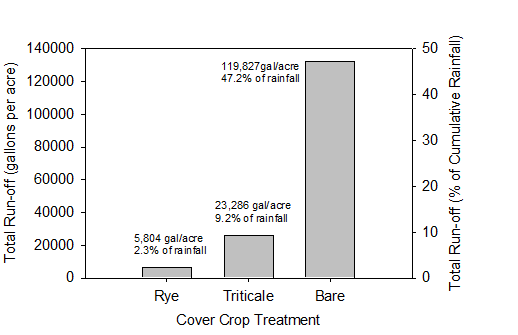
Winter-dormant triticales (e.g. Trios 888) grow more slowly in the winter, which may reduce the risk of producing too much biomass that could be a problem in preparing the soil for the subsequent cash crop (see photos below). Low residue cereal cover crops are typically killed with an herbicide such as glyphosate or a grass selective material such as clethodim or sethoxydim when 60 days old; cereal cover crops on the furrow bottom are difficult to kill mechanically. At this stage the cover crop residue has a low C:N ratio (9-12) and decomposes rapidly in the presence of favorable moisture and temperatures. In studies conducted from 2009 to 2011, we observed that sufficient killed residue remained on the soil surface for 4-6 weeks to continue to protect the surface from raindrop impact and soil crusting. The dead roots of the killed cover crop retain their function of providing channels for rapid infiltration as well. Therefore though the cover crop only grew until mid-January, the soil was protected by the cover crop from about December to mid-March. The cover crop and its residue reduced runoff on a Chualar loam soil where our studies were conducted. We observed that 47.2% of the rainfall ran off of the field in the bare fallow treatment, compared with only 2.3% runoff in the rye and 9.2% in the winter dormant triticale (Figure 5). The rye cover crop increased the amount of water that infiltrated into the soil by 119,827 gallons/A (1/3 of an acre-foot) over the bare fallow treatment. The increased infiltration in the low residue rye cover crop treatment increased the quantity of chloride and sodium leached from the soil by >80% over the bare fallow treatment. This salt leaching provides considerable agronomic benefit, as low rainfall years allow salts to reach levels that may damage crops.
Figure 4. Winter dormant triticale (Trios 102) planted on the furrow bottom. Photo on right is 3 weeks after being treated with glyphosate. Note dead residue covers furrow bottoms.
Figure 5. 2009-2010 Trial. Total runoff from cover crop and bare treatments between mid January and March 7, 2010.
Weed control in the low residue cover crops can be carried out by lillistoning the bed tops and sides, but the furrow sweeps must be lifted to avoid disturbing the protective cover crop residue. You can see an example field preparation following a low residue cover crop in this video https://www.youtube.com/watch?v=k0oVVJ_BA7s . Initial studies with this technique were done with vegetables. There is a delicate balance of covering the soil and obtaining the benefits of increased water infiltration, but having the residue decompose quick enough to allow for ease of bed preparation for planting the subsequent vegetable crop. The quantity of cover crop residue at the time of killing and the spacing of the rain events that allow for cover crop decomposition determines how successfully this technique works. To be on the safe side, in your first efforts using this technique, it is prudent to only use this technique on fields that are scheduled for planting later in the spring to make sure the residue does not create issues for the subsequent cash crop (contact Richard for specific questions: 831-759-7357).
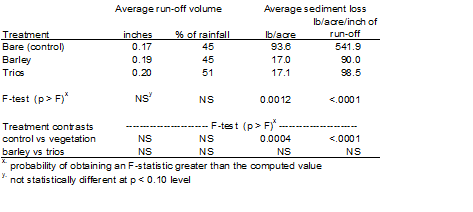
Low Residue Cover crops in Strawberries: Low residue cover crops can also be used in strawberry production systems during the winter. This technique has been used to some extent by growers on hills, and can greatly reduce erosion and improve the water quality of run-off (Table 2). However, cover crops in furrow bottoms were less effective in reducing the quantity of runoff from strawberry fields than in listed vegetable beds. The lack of increased infiltration is due to the volume of runoff generated by beds covered with plastic; soils on the furrow bottom quickly become thoroughly saturated and water quickly runs off. This may be particularly problematic on steeper ground where runoff from individual furrow drainage joins at the end of the rows and becomes an extremely erosive concentrated flow. In this setting a particularly dense seeded cover crop, particularly at the end of rows, may help dissipate the energy of the concentrated flow moving downslope. Often it is necessary to address the lack of improved in-field infiltration by use of underground outlets and sediment basins where runoff carries high sediment loads. Where sediment loads are minimal, vegetated ditches may be helpful to slow the water and allow for infiltration in the ditch (see below).
The use of furrow-bottom cover crops in strawberry systems is still very useful on the east side of the Salinas Valley and other locations on slopes because of reductions in sediment loss. Cover crop choice is important in strawberry systems. For example, fast growing cover crops like barley require more management early in the winter. In our studies, we found that by late December, barley was tall enough to begin shading the strawberry plants. At that point, it can be managed by weed wacking or treatment with a grass selective herbicide like sethoxydim or clethodim (selective for killing grass and do not damage strawberries). In contrast to barley, winter-dormant triticale (Trios 102) grows more slowly and therefore doesn't require management until about late January.
Table 2. Run-off and sediment loss during rain events in a strawberry trial, 2006-07. Data were collected from 11 storm events totaling 4.28 inches.
Low Residue Cover crops: Organic Systems: Low residue cover crops can be used in organic fields as long as they can be killed before they produce too much biomass. Grass cover crops (i.e. cereals) are not recommended, because they are difficult to kill with tillage. Organic herbicides work better on young plants (i.e., cotyledon stage) and are weak on grasses. Growing mustard in the strawberry furrows is one exciting approach that seems to have lots of potential in this region. We've evaluated this over several years at the USDA-ARS in Salinas and have been impressed because mustard establishes quickly with relatively little moisture and is easy to kill with a single cut with a weed whacker. We used hand pushed planters like the ‘Clean Seeder AP' to plant a single line of mustard in the furrow bottom in early December, and typically weed wacked it in late January to early February when it was about the height of the strawberry bed top. Unlike grass cover crops, mustard does regrow after its cut down. After weed whacking, the high-nitrogen mustard residue decomposes quickly and creates relatively few challenges when the furrows are shallow-cultivated to prepare them for strawberry harvesting. Planting a single line in the furrow center helps to keep the base of the mustard plants away from the plastic, which minimizes damage to the plastic during weed wacking. Several mustard cover crop varieties (i.e. Kodiak, Ida Gold, and Caliente) can work, although Ida Gold seems especially well-suited to this system. It grows tall and fills in the furrow quickly, and its relatively large seeds seem more tolerant to deeper planting depths. An appropriate seeding rate for Ida Gold mustard in strawberry furrows is about 10 pounds/acre (approximately $30/acre for seed) which will result in about 30 to 40 plants per foot of furrow bottom. To reduce labor costs with planting mustard, the USDA-ARS has developed a simple planter that plants two strawberry furrows at a time, and will be available for growers to borrow to try. The planter will be demonstrated during a field day at the USDA-ARS in September, and about 1000 pounds of free mustard seed will be available for interested growers to try. Although we have not measured runoff from mustard furrows, we have observed that it dries down furrows and therefore will likely reduce run-off and sediment loss. More research is needed to document the effect of mustard on infiltration and ground water recharge when planted in strawberry furrows, but observation in work done thus far suggests such plantings may be beneficial.
Figure 6. Ida Gold mustard ready for weed whacking, January 30, 2014.
Other In-Field Practices to Increase Infiltration:
- Surface application of gypsum can improve aggregation of soil particles, which improves soil structure, reduces crusting and thereby increases water infiltration into the soil. Gypsum provides calcium which changes the manner in which soil particles are able to flocculate (come together) as aggregates. This approach can be particularly effective for soils that crust, such as those found on the east side of the Salinas Valley. To be effective, gypsum needs to be applied on the surface of the soil (not incorporated) before the first rainfall. The effect of the gypsum declines over time as the rainfall solubilizes the gypsum and carries it away from the soil surface where it is needed.
- Leaving fields unlisted: For fields that will be planted in the late spring, leaving them flat (unlisted) through the winter may also help infiltrate more of the rainfall, as unlisted fields will reduce concentrated flow in furrows where soil may become saturated. If it is possible to leave fields unlisted, leaving a rough soil surface will also help infiltrate a greater portion of the rainfall by creating less runoff potential.
- Tillage can improve water infiltration by breaking the soil crust and slowing runoff by creating a torturous path for the water to follow. Obviously, obtaining access to the field with tillage equipment can be difficult to impossible in wet years.
- 80-inch wide beds appear to have less runoff than 40-inch wide beds in moderate rain storms. However, it is unclear how well 80-inch beds will reduce runoff in a large El Niño rain event of 3 or more inches.