- Contributor: Richard Smith
Bragada bug has now been found in the Santa Maria Valley and there is a strong possibility that it will find its way the Salinas Valley sometime soon. It will be a troublesome insect issue for all Brassica crops as well as peppers. It will be able to establish populations in the many mustard family weeds that occupy areas throughout our valley. These locations will provide a source for infestations of this insect. The following article was written by John Palumbo with the University of Arizona and provides practical information on the means that growers and PCA’s in the desert are using to control this insect. We will be filling our entomology position in December and will have a good resource person to help us deal with this and other insect issues in the Salinas Valley.
Richard Smith.
Please see attached file.
Bagrad Bug 2010 Veg IPM Update
- Author: Steven T. Koike
--Save the Date!--
Food Safety Seminar
Thursday, September 6, 2012
1:00 p.m. to 4:00 p.m.
Sponsored by University of California / California Leafy Greens Research Board
Location: County of Monterey Agricultural Center, Conference Room, 1432 Abbott Street, Salinas
This seminar will present updates and summaries on food safety research, recent food safety issues, and related topics. Speaker: Trevor Suslow, UC Davis.
For more information, the local contact person is Mary Zischke (CLGRB, 831-424-3782).
- Author: Richard Smith
The development of improved cultivation technology for row crop production has been an active area of research, and has made significant progress in recent years. Currently, standard cultivation removes weeds from the majority of the bed using sweeps, knives, coulters and blades. Typically a 4-inch wide band is left around the seedline. Weeds in the uncultivated band are typically removed by hand, and the density of weeds that occur there, determines how laborious and costly subsequent hand weeding will be.
There is technology to remove weeds from the seedline and it generally falls into two categories: 1) blind cultivation and 2) computer assisted cultivators. Implements used for blind cultivation are not guided by a person or machine. They are designed to be able to contact crop plants without significant damage, but yet remove young weed seedlings. Examples of this type of technology include 1) finger weeders, 2) torsion weeders and 3) various spring tine weeders (Photos 1-3). Finger and torsion weeders are useful in row crop scenarios and are designed for use on transplanted vegetables or large seeded vegetables such as beans and corn. Spring tine weeders are mostly used in field crops such as small grains. These devices are designed to take advantage of a size differential between the transplanted crop and newly emerged weeds. Cultivation is often done 1-2 weeks following transplanting. In our studies, these weeders supplement the action of standard cultivation and have the ability to reduce the time it takes to subsequently hand weed vegetables (Table 1).
Camera and computer assisted weeders are beginning to become available on the market. This technology can detect and distinguish crop plants in the seedline. An image of the seed row is captured by the camera and is then analyzed by the computer, which determines the location of crop plants in the seedline. Current technology uses the difference in size and color between the crop and the weeds, to distinguish the crop from weeds. These mechanical weed control machines were designed primarily for use on transplanted crops because the crop plants are initially larger than the weeds. Once the machine recognizes the crop plants it activates a weed removal implement. The implements that have been proposed or that are used by commercial units include swinging, spinning, or opening and closing blades, or other techniques such as flaming and the use of timed chemical sprays. The key for success is for the machine to take out as many weeds in the seedline as possible while not damaging crop plants. There has been more research and adoption of this technology in Europe; however, in California, there are two notable computer-assisted mechanical weed control machines either on the market or close to being commercialized for row crop production:
The Tillet Weeder, fabricated in England (Garford Corp, http://garford.com/) uses camera and computer technology to direct spinning round-blade with a notch cut out of one side. The blade travels in the seed line removing weeds, but when it encounters a crop plant, it spins around it by placing the plant in the notch (Photos 4 & 5). We evaluated this machine from 2009-2010 and found it to be effective in removing weeds from the seedline and reducing subsequent hand weeding time (http://cemonterey.ucdavis.edu/Vegetable_Crops/Weed_Reports/ for more info).
A machine is being developed by the University of Arizona and Mule Deer Automation (New Mexico) that sprays a chemical in a band application to remove unwanted plants. Various chemicals can be used in this machine such as acid or salt-based fertilizers (e.g., phosphoric acid or ammonium nitrate) or herbicides such as paraquat, pelargonic acid (Scythe®), or organic herbicides (Photo 6).
Other technologies that are being developed include alternative detection systems such as the use of lasers or x-rays to detect the stem of crops such as tomatoes. The development of plant maps by use of real time kinematic (RTK) global positioning systems (GPS) is being used in field crops. This technology allows for closer and more precise guidance of standard cultivators. This technology has not been applied to vegetable production to any extent in our area, but is being researched as a technique to someday guide in-seedline cultivators.
Technology will continue to develop and improve in the coming years. Computer-assisted mechanical weed control machines can provide an alternative option for weed control in vegetable crop production. These technologies do not entirely replace the need for hand labor, but they can make subsequent hand weeding operations less costly and more efficient.
Photos 1-3. Blind cultivation weeders: Finger weeders - left, torsion weeders - middle and spring tine weeder – right
Photos 4 & 5. The Tillet Weeder is a commercially available mechanical weed control machine that uses computer technology and a spinning blade to remove weeds. Left photo: The disc-shaped cultivation blades are lifted up so you can observe the notched cut-out that allows the blade to spin around crop plants. Right photo: Thinning lettuce.
Photo 6. Lettuce thinned and weeded with the University of Arizona/Mule Deer Automation prototype. The unwanted plants were treated with ammonium nitrate (AN20) fertilizer (in dark gray rectangular areas) and will die in a matter of days.
Table 1. 2010 finger weeder trial. Weed removal by cultivation treatments, weeding time and yield evaluation (count of heads remaining in field after harvest operation)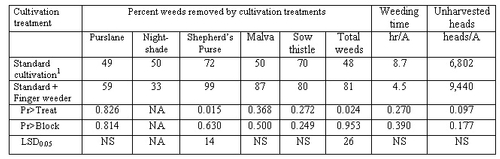
For further information on automated cultivators for vegetable crops:
Blind Cultivators
- YouTube video on blind cultivation techniques developed by UCCE, Monterey County: http://www.youtube.com/watch?v=I4kzebMG6rE
- Buddingh Weeder Co. (finger weeder and others) http://www.buddinghweeder.com/
- Einboch (spring tine weeder) http://www.einboeck.at/index.php?lang=en
- Frato Machine Import (torsion weeder) http://www.frato.nl/UK/torsiewieder-UK.htm
- Kovar (spring tine weeder) http://www.kovarsales.com/weederharrow.html
- Kress Company (finger weeder) http://www.kress-landtechnik.de/
- Red Dragon (flame weeder) http://www.flameengineering.com/Agricultural_Flamers.html
Computer Assisted Cultivators
- Garford Agricultural Equipment http://www.garford.com/index.html
- Frank Poulsen Engineering http://www.visionweeding.com/Products/Intra%20Row%20Weeding/ROBOVATOR.htm
- Ramsey Highlander http://www.ramsayhighlander.com/products/spinach-spring/spinach.htm
- Steketee Corporation (Netherlands): http://www.steketee.com/category/Mechanische-onkruidbestrijding (also see YouTube of their implement: http://www.youtube.com/watch?v=y7PJWwAAo1U
- University of Arizona (Mark Siemens) http://extension.arizona.edu/programs/specialty-crops-mechanization
- University of California, Davis (David Slaughter) http://bae.engineering.ucdavis.edu/pages/faculty/slaughter.html and http://baesil.engineering.ucdavis.edu/BAESIL/AutoWeedControl.html (videos of the implement)
- Author: Richard Smith
- Author: Michael D Cahn
Conversion between nitrate (NO3) and nitrate-nitrogen (NO3-N):
|
To convert |
To |
Multiply by |
|
Nitrate (NO3) |
Nitrate-nitrogen (NO3-N) |
0.22 |
|
Nitrate-nitrogen (NO3-N) |
Nitrate (NO3) |
4.43 |
The reason for this conversion is that nitrate molecule weighs 62 grams per mole; the nitrogen content of nitrate is 22.5% of the total weight of the molecule.
Nitrogen content of irrigation water*
|
Water content of |
Multiply by |
To determine |
|
PPM NO3 |
0.052 |
Pounds N/acre inch |
|
PPM NO3 |
0.62 |
Pounds N/acre foot |
|
PPM N03-N |
0.23 |
Pounds N/acre inch |
|
PPM N03-N |
2.74 |
Pounds N/acre foot |
* water analyses from most labs report NO3 in units of ppm, but it is very important to pay attention to which units the results are reported.
How much of the nitrogen in water should be credited to your crop is debatable. Consider that lettuce transpires 5 to 8 inches of water between germination and maturity in the Salinas Valley during the summer. Extra water applied beyond crop ET would be lost as drainage and therefore would not contribute N to the crop. The extra water also would likely leach plant available soil nitrate below the root zone. In addition, some ground water that is high in nitrate is also high in salts and may require a leaching fraction (extra water applied to leach salts below root zone) to attain maximum production. The good news is that you can account for the N contribution from the nitrate in the irrigation water using the quick nitrate soil test for previous irrigations. However, this test will not estimate the contribution of N from the irrigation water for future waterings.
Our best estimate of how much N the irrigation water would contribute to future waterings is to divide the crop evapotranspiration by the irrigation efficiency. For example, for 7 inches of crop ET and an 80% irrigation efficiency, the following values would approximate the N contribution of irrigation water for the indicated range of nitrate concentrations:
|
Nitrate (NO3) concentration in irrigation water |
Nitrate (NO3-N) concentration in irrigation water |
Lbs nitrogen/A in seven inches of irrigation water taken up by lettuce* |
|
45 |
10 |
13 |
|
89 |
20 |
25 |
|
177 |
40 |
51 |
|
266 |
60 |
76 |
* multiplied by 0.8 to account for the irrigation system efficiency
As can be seen, waters containing less than 45 ppm NO3 generally do not contribute a significant amount of nitrogen for crop growth. However, if well waters contain more than that amount they begin to contribute greater amounts of water for crop growth.
- Author: Richard Smith
- Author: Steven T. Koike
In the last year we have been called out to look at lettuce fields showing uneven growth. The fields looked like they were not growing normally due to a cultural practice. The lettuce plants in the field vary in size with normal plants interspersed with stunted plants (Photos 1 and 2). The patterns in the field do not fit with sprinkler patterns or mechanical issues because affected plants are more randomly dispersed. The pattern may fit with soil insect issues, but no soil insects were present in the affected fields. Sometimes stunted plants were more prevalent in areas that might have poor drainage. The randomness of affected plants is similar to some of the nitrogen toxicity issues that we have detected in some fields (http://ucanr.org/blogs/blogcore/postdetail.cfm?postnum=4931). In some cases the taproot of the plant is missing (a typical symptom of nitrogen toxicity), but taproots are not consistently missing and therefore the symptoms do not fit nitrogen toxicity. Upon close examination of the root system, small necrotic sections of the root are present and we were able to confirm that the lettuce was affected by black root rot disease.
Root symptoms of black root rot of lettuce consist of numerous dark brown to black bands and lesions on small feeder and larger secondary roots (Photos 3 and 4). Internal vascular discoloration (as seen with Verticillium wilt) is absent. If severe infections occur early in the crop cycle, the lettuce seedlings will have damaged taproots and experience delayed growth, resulting in stunted, small plants that otherwise may look healthy. The stunting is usually most evident from rosette stage through crop maturity. In advanced cases, the lower leaves of the plant may turn yellow or even brown. In the Salinas Valley, black root rot has been seen to cause stunting and resulting crop loss on both iceberg and romaine cultivars. Black root rot of lettuce has been known for many years in Australia and other regions. The disease was first documented on lettuce in California in 2005. Our diagnostic lab has periodically received black root rot cases since then.
Black root rot is caused by the soilborne fungus Thielaviopsis basicola. The fungus makes two types of spores, with one type being a thick walled spore (Photo 5) that resists weathering and enables the fungus to survive for long periods in the soil. This pathogen is reported to have a broad host range that includes the following vegetable crops: bean, beet, carrot, garlic, lettuce, onion, pea, pepper, tomato. In coastal California we have not found black root rot on most of these crops; black root rot disease has been confirmed only for lettuce and tomato.
Differences in strains or isolates of T. basicola may exist. For example, researchers found that when inoculated onto lettuce and bean, T. basicola isolates from lettuce were very aggressive and caused significant damage. However, the same lettuce isolates caused very little damage to other known hosts. Such research indicates that T. basicola may consist of isolates having some host preferences.
Industry-wide, black root rot of lettuce is a very minor problem that apparently only affects a few fields. However, growers and PCAs should be aware of this disease and notify Farm Advisors if black root rot becomes more common. For other crops, black root rot is managed by a combination of resistant cultivars, crop rotation to non-hosts, sanitation practices so as to not spread contaminated soil/mud to uninfested fields, and application of fungicides to seedlings.

Photo 1. Typical pattern of Thielaviopsis basicola infection in lettuce.

Photo 2. Stunted lettuce affected with black root rot (on left)

Photo 3: Diseased roots of lettuce affected by black root rot.

Photo 4. Close-up of black root rot lesion on fine feeder roots of lettuce.
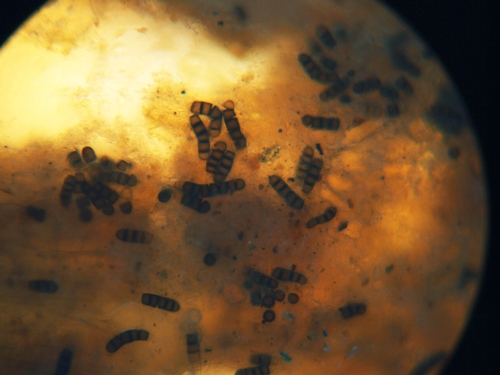
Photo 5: Dark conidia of Thielaviopsis basicola forming on lettuce root.





