- Author: Steven T. Koike
Beginning in early April, 2012, the UC Cooperative Extension diagnostic lab in Salinas began to receive lettuce samples exhibiting obvious symptoms of a virus problem. Samples continued to be submitted throughout the month. All samples tested positive for the thrips-vectored Impatiens necrotic spot virus (INSV). This virus first began to cause damage to California lettuce in 2006. Since that time, INSV has occurred to a greater or lesser degree every season. However, significant INSV outbreaks usually are found beginning in late May or early June. Seeing INSV on lettuce in April is therefore unusual and growers and pest control advisors should carefully monitor these situations.
INSV-infected plants have leaves with brown to dark brown spots and dead (necrotic) areas; this necrotic tissue can resemble burn damage caused by pesticide or fertilizer applications (see photos below). Affected leaves can be distorted and twisted. Extensive necrosis can cause much of the leaf to become brown, dry, and dead. Some leaf yellowing can also be observed. Yellowing and the brown spotting tend to be observed on the newer leaves near the center of the plant’s growing point. If plants are affected with INSV early in their development, growth may be stunted. All lettuce types are susceptible, and INSV has been confirmed on iceberg, butterhead, romaine, and leaf lettuces. INSV can infect many other crops and weed species; the virus is vectored by the Western flower thrips (Frankliniella occidentalis).
Growers, PCAs, and other field personnel should exercise caution in diagnosing INSV disease because the lettuce dieback viruses cause very similar symptoms (see table below). Lettuce dieback is caused by two pathogens: Lettuce necrotic stunt virus (LNSV) and Tomato bushy stunt virus (TBSV). Infected lettuce can be severely stunted, especially if infected early in plant development. The oldest, outer leaves can be severely yellowed. Brown, necrotic spots and lesions later develop in these outer leaves. The younger, inner leaves remain dark green in color, but can be rough and leathery. LNSV/TBSV infects only romaine, butterhead, and leaf lettuces; modern cultivars of iceberg lettuce are immune. The LNSV/TBSV virus complex is a soilborne problem and no vector (insect, nematode, fungus) is known to spread these viruses.
For help in diagnosing these and other plant problems, submit samples to the UC Cooperative Extension diagnostic lab in Salinas. 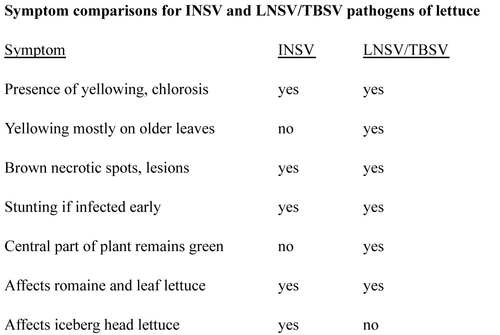
Photos: Impatiens necrotic spot virus (INSV) on lettuce.






- Contributor: Steven Koike
- Author: Steven T. Koike
Weather and lettuce diseases. Late spring rains, cold temperatures, and high humidity are making it possible for two important foliar diseases of lettuce to show up in California and cause damage in 2012. Both bacterial leaf spot and anthracnose have been observed in numerous fields throughout the central coast. If rains continue or if the crop is irrigated with sprinklers, both diseases can result in significant damage to lettuce leaves and resulting loss of yields due to poor quality of the harvested heads. Experienced field personnel will likely recognize these two problems without difficulty; however, we recommend that if there is any question, laboratory tests be conducted to confirm diagnosis.
Bacterial leaf spot. Initial symptoms are small (1/8 to 1/4 inch), water-soaked spots that occur on the older, outer leaves of the plant. Lesions are typically angular in shape and quickly turn black (photo 1)—this is the diagnostic feature of this disease and readily separates this from anthracnose (which causes white to light pink lesions). If disease is severe, numerous lesions may coalesce, resulting in the collapse of the leaf. Older lesions dry up and become papery in texture, but retain the black color. Lesions rarely occur on newly developing leaves.
Bacterial leaf spot is caused by bacterium Xanthomonas campestris pv. vitians. The pathogen is highly dependent on wet, cool conditions for infection and disease development. Splashing water from overhead irrigation and rain disperses the pathogen in the field and enables the pathogen to infect significant numbers of plants. The pathogen can be seedborne and is introduced into the field via contaminated seed. In addition, the bacterium can survive for up to five months in the soil. Therefore, infected lettuce crops, once disked into the soil, can supply bacterial inoculum that can infect a subsequent lettuce planting.
Spray applications are not effective at managing bacterial leaf spot of lettuce. The disease is managed by using uninfested seed, irrigating the crop via furrow or drip, and avoiding back-to-back lettuce rotations if the first crop was diseased.
Anthracnose. Early symptoms are small (1/8 to 1/4 inch), water-soaked spots occurring on outer leaves. Spots enlarge, turn yellow then tan, and are usually angular in shape. Under cool, rainy conditions, white to pink spore masses of the fungus will be visible in the centers of the tan colored lesions (photo 2)—this is the diagnostic feature of this disease and readily separates this from bacterial leaf spot (which causes black lesions). If disease is severe, the lesions will coalesce and cause significant dieback of the leaf and in some cases will result in stunting of the plant. As spots age, the affected tissue will dry up and become papery in texture. Eventually the centers of these spots can fall out, resulting in a shot hole appearance. Anthracnose lesions are often clustered along the midribs of lower leaves.
Anthracnose is caused by the fungus Microdochium panattonianum. This fungus infects only lettuce and does not cause disease on any other crop. The fungus can survive for up to four years as microsclerotia in soil. The anthracnose pathogen requires cool, wet conditions for infection and symptom development and hence is associated with rainy weather. Splashing water moves microsclerotia and conidia from soil onto leaves, resulting in infection.
To manage anthracnose, first avoid planting lettuce in fields having a history of the disease. Use irrigation systems (furrow or drip irrigation) that reduce or eliminate splashing water and leaf wetting. Apply protectant fungicides, such as strobilurins, which are effective for controlling this disease.

Photo 1. Bacterial leaf spot results in black, angular shaped lesions on lettuce leaves.
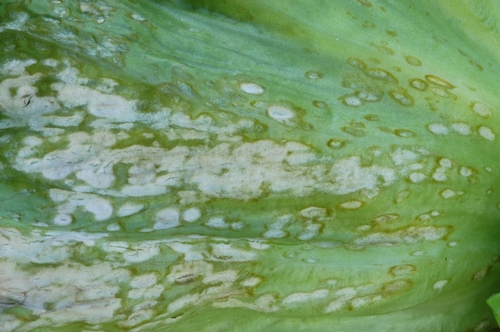
Photo 2. On lettuce, anthracnose results in tan, angular shaped lesions that are covered with the white to pink growth of the fungus.
- Author: Richard Smith

A video entitled, "Alternative Cultivators for Organic Vegetable Production" is now available on YouTube. The video was produced by Aaron Heinrich and Richard Smith and presents information on new developments in automated weed control technology and research information on blind cultivators such as torsion and finger weeders. The video can be accessed at:
http://www.youtube.com/watch?v=I4kzebMG6rE
For more information on alternative cultivation see the blog below. Also, go to
http://cemonterey.ucdavis.edu/Vegetable_Crops/Weed_Reports/
for research reports on precision cultivation.
- Author: Richard Smith
The development of improved cultivation technology for row crop production has been an active area of research, and has made significant progress in recent years. Currently, standard cultivation removes weeds from the majority of the bed using sweeps, knives, coulters and blades. Typically a 4-inch wide band is left around the seedline. Weeds in the uncultivated band are typically removed by hand, and the density of weeds that occur there, determines how laborious and costly subsequent hand weeding will be.
There is technology to remove weeds from the seedline and it generally falls into two categories: 1) blind cultivation and 2) computer assisted cultivators. Implements used for blind cultivation are not guided by a person or machine. They are designed to be able to contact crop plants without significant damage, but yet remove young weed seedlings. Examples of this type of technology include 1) finger weeders, 2) torsion weeders and 3) various spring tine weeders (Photos 1-3). Finger and torsion weeders are useful in row crop scenarios and are designed for use on transplanted vegetables or large seeded vegetables such as beans and corn. Spring tine weeders are mostly used in field crops such as small grains. These devices are designed to take advantage of a size differential between the transplanted crop and newly emerged weeds. Cultivation is often done 1-2 weeks following transplanting. In our studies, these weeders supplement the action of standard cultivation and have the ability to reduce the time it takes to subsequently hand weed vegetables (Table 1).
Camera and computer assisted weeders are beginning to become available on the market. This technology can detect and distinguish crop plants in the seedline. An image of the seed row is captured by the camera and is then analyzed by the computer, which determines the location of crop plants in the seedline. Current technology uses the difference in size and color between the crop and the weeds, to distinguish the crop from weeds. These mechanical weed control machines were designed primarily for use on transplanted crops because the crop plants are initially larger than the weeds. Once the machine recognizes the crop plants it activates a weed removal implement. The implements that have been proposed or that are used by commercial units include swinging, spinning, or opening and closing blades, or other techniques such as flaming and the use of timed chemical sprays. The key for success is for the machine to take out as many weeds in the seedline as possible while not damaging crop plants. There has been more research and adoption of this technology in Europe; however, in California, there are two notable computer-assisted mechanical weed control machines either on the market or close to being commercialized for row crop production:
The Tillet Weeder, fabricated in England (Garford Corp, http://garford.com/) uses camera and computer technology to direct spinning round-blade with a notch cut out of one side. The blade travels in the seed line removing weeds, but when it encounters a crop plant, it spins around it by placing the plant in the notch (Photos 4 & 5). We evaluated this machine from 2009-2010 and found it to be effective in removing weeds from the seedline and reducing subsequent hand weeding time (http://cemonterey.ucdavis.edu/Vegetable_Crops/Weed_Reports/ for more info).
A machine is being developed by the University of Arizona and Mule Deer Automation (New Mexico) that sprays a chemical in a band application to remove unwanted plants. Various chemicals can be used in this machine such as acid or salt-based fertilizers (e.g., phosphoric acid or ammonium nitrate) or herbicides such as paraquat, pelargonic acid (Scythe®), or organic herbicides (Photo 6).
Other technologies that are being developed include alternative detection systems such as the use of lasers or x-rays to detect the stem of crops such as tomatoes. The development of plant maps by use of real time kinematic (RTK) global positioning systems (GPS) is being used in field crops. This technology allows for closer and more precise guidance of standard cultivators. This technology has not been applied to vegetable production to any extent in our area, but is being researched as a technique to someday guide in-seedline cultivators.
Technology will continue to develop and improve in the coming years. Computer-assisted mechanical weed control machines can provide an alternative option for weed control in vegetable crop production. These technologies do not entirely replace the need for hand labor, but they can make subsequent hand weeding operations less costly and more efficient.



Photo 6. Lettuce thinned and weeded with the University of Arizona/Mule Deer Automation prototype. The unwanted plants were treated with ammonium nitrate (AN20) fertilizer (in dark gray rectangular areas) and will die in a matter of days.
For further information on automated cultivators for vegetable crops:
Blind Cultivators
- YouTube video on blind cultivation techniques developed by UCCE, Monterey County: http://www.youtube.com/watch?v=I4kzebMG6rE
- Buddingh Weeder Co. (finger weeder and others) http://www.buddinghweeder.com/
- Einboch (spring tine weeder) http://www.einboeck.at/index.php?lang=en
- Frato Machine Import (torsion weeder) http://www.frato.nl/UK/torsiewieder-UK.htm
- Kovar (spring tine weeder) http://www.kovarsales.com/weederharrow.html
- Kress Company (finger weeder) http://www.kress-landtechnik.de/
- Red Dragon (flame weeder) http://www.flameengineering.com/Agricultural_Flamers.html
Computer Assisted Cultivators
- Garford Agricultural Equipment http://www.garford.com/index.html
- Frank Poulsen Engineering http://www.visionweeding.com/Products/Intra%20Row%20Weeding/ROBOVATOR.htm
- Ramsey Highlander http://www.ramsayhighlander.com/products/spinach-spring/spinach.htm
- University of Arizona (Mark Siemens) http://extension.arizona.edu/programs/specialty-crops-mechanization
- University of California, Davis (David Slaughter) http://bae.engineering.ucdavis.edu/pages/faculty/slaughter.html and
http://baesil.engineering.ucdavis.edu/BAESIL/AutoWeedControl.html (videos of the implement)
Table 1. 2010 finger weeder trial. Weed removal by cultivation treatments, weeding time and yield evaluation (count of heads remaining in field after harvest operation)
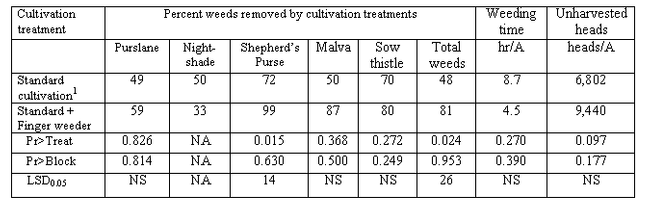
1 – Standard cultivation leaves a 4 inch wide uncultivated band around the seedline




