- Author: Alejandro Del Pozo-Valdivia
Since last week, I have been receiving samples with “red” aphids to get the identification. It turned out that this time, most of these ‘reddish' aphids were identified as the lettuce aphid, Nasonovia ribis-nigri (Fig. 1). To me, they look more red-orange; however, their distinct black marks on the abdomen and short cauda (finger-like, short appendage at the end of the abdomen) are some key ID features. These features help to differentiate the lettuce aphid from the other “red” aphid, the potato aphid Macrosiphum euphorbiae (Fig. 2). Yes, we do have two different species of red aphids!
More samples for aphid ID are still coming into the UC Cooperative Extension office. The pattern is still similar to last week. There are mostly lettuce aphids on the submitted samples. I was also able to notice that some samples have mixed populations between the lettuce and the potato aphids, where all the specimens were red.
We have several trial locations where we are scouting for aphids. So far, fields in Soledad have the largest number of aphids documented, as both alates (with wings, collected from yellow sticky cards) and wingless (collected from lettuce samples). If you need further information about the other scouting locations, or would like to double check your aphid ID, please contact or send samples to Alejandro Del-Pozo (adelpozo@ucanr.edu, 831-759-7359).
- Author: Alejandro Del Pozo-Valdivia
I have been receiving phone calls about the presence of “red aphids” in organic romaine lettuce that is resistant to the lettuce aphid, Nasonovia ribis-nigri (Hemiptera: Aphididae, Fig. 1). The lettuce aphid is considered a difficult pest to manage. This aphid can be green as well as red, and will be usually found in large colonies inside the heart of the lettuce head. Being inside the lettuce head protects these aphids from predators and parasitoids; and their presence is considered a contaminant when heads are harvested.

However, none of the samples that were submitted to the UC Cooperative Extension diagnostic laboratory were identified as the lettuce aphid. What are those “red aphids”?
First of all, aphids could have different color morphs within the same species. Therefore, relying only on color to identify aphids is not the ideal character to tell apart these pest species.
Submitted “red” aphid samples were identified as either: 1) the potato aphid, Macrosiphum euphorbiae, and 2) the foxglove aphid, Aulacorthum solani. Potato aphid has both the green and the red color morphs (Fig. 2). On the other hand, the foxglove aphid is usually green (Fig. 3); but infections of a naturally occurring Entomophthora fungus will make the specimens look like reddish. This sort of red coloration is due to the sporulation of the fungus from the dead aphid stuck on the leaves (Fig. 4).



If you are managing organic lettuce with resistance to the lettuce aphid and you find red aphids, it is important to consider other features beside color. Additional information on how to identify aphid species infesting lettuce can be found at http://ipm.ucanr.edu/PMG/selectnewpest.lettuce.html. If you would like to have a second opinion on your identification, you can always call and/or send your sample to the UC Cooperative Extension office in Salinas.
- Author: Shimat Villanassery Joseph
Springtail (Protaphorura fimata) (Figure 1) is a serious pest of lettuce in the northern part of Salinas Valley of California. The direct seeded young lettuce seedlings in fields with high densities of springtail show retarded or stunted growth and do not emerge in a synchronous pattern (Figure 2). Springtails are reported to feed on soil fungi, decaying plant materials and live roots.
Springtails attack the germinating seeds of the lettuce, but it is not certain if irregular or inconsistent plant stand is due to the persistent feeding by springtail on both germinating and developing seedling stages of lettuce. Springtail continue to occur in the soil beyond 30 days after planting. Knowing the most vulnerable stage(s) of lettuce to springtail feeding will help in determining the best timing for control measure intervention to achieve a uniform lettuce stand.
Lettuce fields are heavily irrigated at least once before and up to three weeks after planting the seeds for uniform seed germination and seedling establishment. However, the behavioral response of springtail to feeding injury on lettuce under high soil moisture condition has not yet been studied in the central coast of California.
Similarly, the temperature has a profound impact on lettuce plant development as well as the growth and activities of springtail. Springtail has been found causing crop losses during February to May in the Salinas Valley and beyond June, springtail related problems are not widely reported. Perhaps slower lettuce seed germination and subsequent development during cooler seasons (January to May) is the pre-disposing factor as seedlings are exposed to springtails for an extended time frame than during the rest of the year. The relationship between temperature and springtail feeding of germinating lettuce seeds has not been investigated.
A study was conducted to determine the effect of germinating stages of lettuce seeds (up to 7 days after planting), soil temperature and moisture to springtail feeding injury.
Germinating seeds or one day old lettuce seedlings were the most vulnerable stage to springtail feeding, resulting in reduction in seedling growth. Thus, it appears that once the roots are established in the soil, lettuce is less susceptible to springtail feeding injury. Because the germinating phase of the plants is more likely to be injured, springtail monitoring activity should start prior to planting the seeds to determine the presence of springtail in the soil. Previous studies showed that, beet or potato slice baits attract springtail if placed in the top layer of the soil; thus, these baits could be used for monitoring springtail activity in the soil. If the soil is not moist, the baits may not capture springtail and springtail activity may go undetected.
When the experiments were conducted with germination phase in the temperatures as low as 41°F, springtail feeding was still evident. This suggests that although the seed germination and seedling development is progressing slowly in the cooler temperatures, springtail can be still active in feeding if there is sufficient moisture in the soil. Also, this suggests that lettuce seedlings might require prolonged protection from springtail with additional insecticide sprays until the seedlings are established in the cooler temperatures especially in spring and early summer (January to May). In the later part of summer and fall, the temperatures are higher than 60°F even at nights, which allows the seeds to germinate and develop quickly and not providing springtail to persistently feed and cause economic damage. In these circumstances, an at-plant application of insecticide is likely to provide adequate springtail control and multiple applications may not be required.
High moisture content in the soil will favor springtail feeding on the germinating lettuce seeds. In the Salinas Valley, before the lettuce seeds are planted, fields are pre-irrigated to aid land preparation and bed shaping. It has been observed that the springtail density increased from the sub-surface of soil when the field was recently irrigated or after a rain event. This cultural practice which maintains high moisture levels for seed germination on the sub-surface profiles of the soil might be favoring the faster buildup of springtail populations. Springtail captures in bait traps were greater immediately after irrigation.
Clearly, this study demonstrates that early lettuce seed development stages are the most vulnerable to springtail feeding injury. Moisture has a profound effect on springtail feeding on germinating lettuce seeds. This study also suggests that springtail can attack the germinating lettuce seeds at all growing temperatures in the Salinas Valley, although the seed germination and subsequent seedling development at cooler temperatures would increase the vulnerability of lettuce seeds to springtail feeding. This information provides insights not only on the timing of protection but the extent of protection under various temperature ranges also in managing springtail in the Salinas Valley. Plants growing the cooler temperature need prolonged protection for springtail if adequate moisture is present in the top soil of the bed. In the warmer temperatures, seed development would occur rather quickly which suggests that prolonged protection against springtail is not necessary. These results warrant the need for more field studies on protecting lettuce seeds from springtail in the cooler temperatures especially during spring and early summer lettuce plantings in the Salinas Valley.
If interested in the details of the study, please read the published article:
http://cemonterey.ucanr.edu/files/253284.pdf
- Author: Shimat Villanassery Joseph
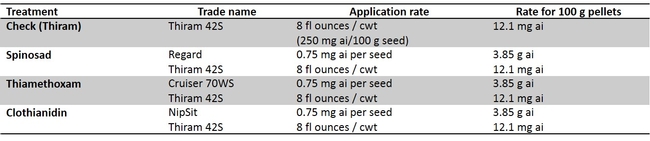
A series of laboratory and field studies were conducted to determine if the insecticides coated lettuce seeds are an option to control key lettuce pests in the Salinas Valley: springtail (Protaphorura fimata; Fig. 1A), leafminers (Liriomyza spp.; Fig. 1B) and western flower thrips (Frankliniella occidentalis; Fig. 1C). In addition, a laboratory test was conducted to determine if “primed” lettuce seeds reduced springtail feeding damage.
Springtails. Springtail (P.fimata) is soil dwelling primitive arthropod primarily attacks germinating lettuce seeds, reducing the plant vigor or death, which cause patchy or area-wide stand loss. Most springtails possess a forked organ (furcula) in the rear-end, which is extended forward and backward to jump; hence, the common name, springtail. However, the springtail species, sampled from lettuce fields causing the stand loss, does not have furcula. This means it cannot jump.
The head lettuce seed ‘Regency' was coated with clothianidin, thiamethoxam, and spinosad (Table 1). The seeds were coated by Dr. Alan Taylor at Cornell University and coating technique mirrored commercial seed coating procedure. Laboratory studies were conducted in containers with springtail (P. fimata) infested soil. The data show that all three insecticides spinosad, clothianidin and thiamethoxam treated seeds significantly reduced the incidence of springtail feeding injury when compared with untreated seeds. Among insecticides, superior performance in efficacy was noted in the following order: clothianidin > thiamethoxam > spinosad (Fig. 2). Two field trials were conducted against springtails using the same seed treatments, however, the springtail pressure was so low that conclusive data were not obtained. Clothianidin (NipSit) in particular, is now registered on head lettuce and could be used for springtail control. This is an important information in that springtails attack the germinating seeds of lettuce especially in the spring time. During spring, we get some rain showers and the wet conditions in the field after planting makes insecticide application along seed line almost impossible. If the insecticide coated seeds are planted, the grower or PCA could avoid at-plant insecticide application which is typically targeted toward springtails. Application of insecticides such as neonicotinoids and pyrethroids along the seed line will protect the germinating seeds from springtail feeding. More field studies will be conducted in the following years to validate these results in the field.
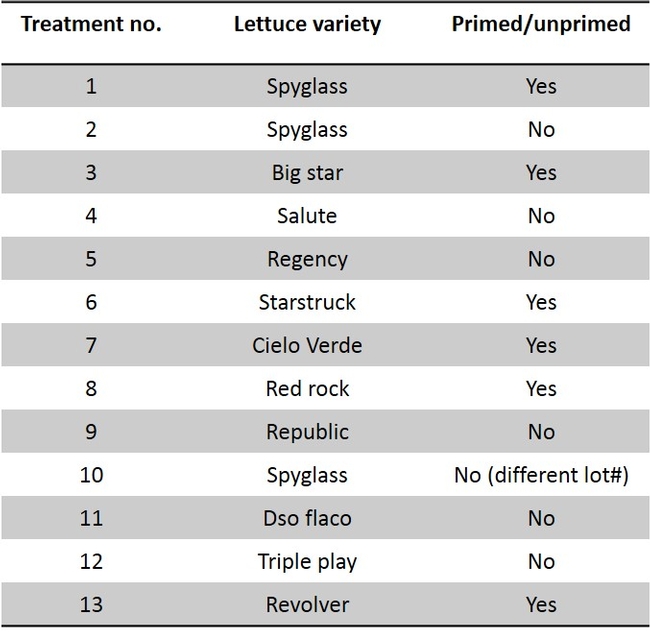
Studies were also conducted to determine if there are any varietal effects exists (Table 2). The much needed attribute for springtail control is faster seed germination so that the springtail would not get sufficient time to feed and cause seed mortality. “Primed” lettuce seeds are used for uniform and a quick germination (cut short 2 to 3 days than “unprimed” seeds). “Primed” and “unprimed” seeds were evaluated to determine if the quick seed development would reduce springtail damage. Data show that germinating “primed” seeds were impacted with springtail feeding affecting their germination and were not different from the “unprimed” seeds when the springtail pressure was moderate to high (Fig. 3). The seeds used for this experiment were from same seed lot (“primed” and “unprimed”) for a lettuce variety. Also, there was no clear variety difference in springtail feeding damage.
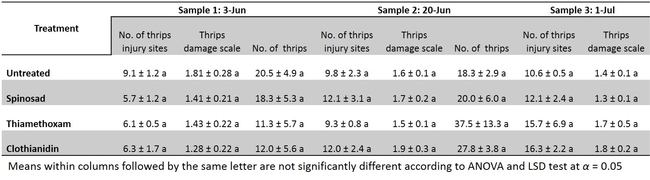
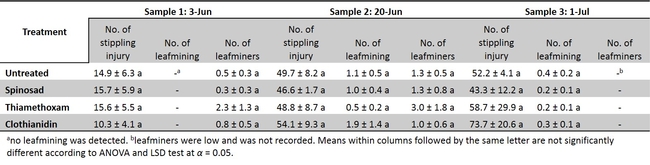
Leafminers and western flower thrips. The leafminer eggs are laid within the surface layer of the leaf. The eggs hatch within couple of days and the maggots mine through the surface layer of the leaves. The egg laying and maggot mining creates stippling and mining injuries which make the leaves unmarketable. Although UC recommends few insecticides such as Agri-mek (Abamectin), Trigard (Cyromazine), Aza-direct (Azadirachtin) and Entrust (Spinosad), the management of leafminers are primarily relied upon on Agri-mek applications.
Thrips is another of the major pest of lettuce, and combination of direct feeding injury and viral disease [thrips-transmitted tospoviruses [Impatiens Necrotic Spot Virus (INSV)] can cause significant losses in lettuce production. In addition, because most of the export markets have set higher standards on prevalence of live and dead thrips in the produce, the lettuce industry is constantly battling ways to significantly reduce thrips in the produce targeted for export.
A replicated-field trial was conducted to determine the efficacy of seed coated insecticides (Table 1) on leafminers and western flower thrips incidence and their infestation. The results show that insecticide seed coating may not be an effective option for leafminers and thrips control in head lettuce (Table 3 and 4) under the conditions this experiment was conducted. There was no reduction of leafminer or thrips feeding with insecticide coated seeds compared with untreated control. Further evaluations under varying conditions might be necessary to validate the consistency of these results.

- Author: Shimat Villanassery Joseph
The soil-dwelling springtail (Protaphorura fimata) is a serious pest of lettuce in the Salinas Valley of California. This springtail is ~ 2.5 mm long and white and has no jumping organ (furcula) and eyes. They feed primarily on the radicle of the germinating lettuce and broccoli seeds causing stunted seedling growth, and their high densities at planting is associated with poor lettuce stands. Most springtails are saprophytic feeders considered as beneficial organisms because they aid in the decomposition of decaying plant material, thereby contributing to the cycling of carbon and nitrogen, which in turn improves soil health and structure. These springtails typically known to feed on fungi in the soil.
There is no grower-friendly monitoring method to determine incidence and abundance of springtails in the lettuce and broccoli fields of California's central coast. Garden symphylan another soil-borne arthropod pest that occurs in vegetable fields, is commonly monitored using a potato slice bait. Potato slices are typically deployed on the soil surface, and they attract garden symphylan. The utility of potato slices for captures of springtail has not been investigated. Because potato slices are off-white in color, it might be difficult to quickly quantify lightly-colored arthropods such as springtail and garden symphylan on the potato surface. Beets are dark red in color and may provide background contrast and to help quantify lightly-colored organisms on it.
Recent research suggest that beet and potato attract springtail when placed in lettuce fields and these baits could be used to monitor in commercial field settings. It could only provide the presence or absence information, which might be still important to avoid unnecessary insecticide application with no or low captures on baits. Capture of springtails on the baits suggests that their populations are likely developing in the upper soil profile especially near the root system. Also, the incidence and abundance of springtail is subjected to soil moisture and their captures likely decline sharply as the upper soil layer loses moisture. It is likely that springtail populations multiply as the pre-plant irrigation is initiated. Monitoring the field using baits before or during the initial irrigation may provide an indication of active springtail population in the upper soil profile. Study also show that greater numbers of springtails were collected on beet one day after deployment than extended days of exposure. Perhaps deployment of baits for extended periods of time in soil may cause desiccation and make them less attractive to springtails.
Therefore, beet and potato can attract springtail in the soil and could be used for monitoring springtail.
If you are interested to read further on the published article, click the link below.
http://cemonterey.ucanr.edu/files/230154.pdf