Authors: Surendra K. Dara, Entomology and Biologicals Advisor, University of California Cooperative Extension
Western flower thrips adult (Photo courtesy: Jack Kelly Clark, UC IPM)
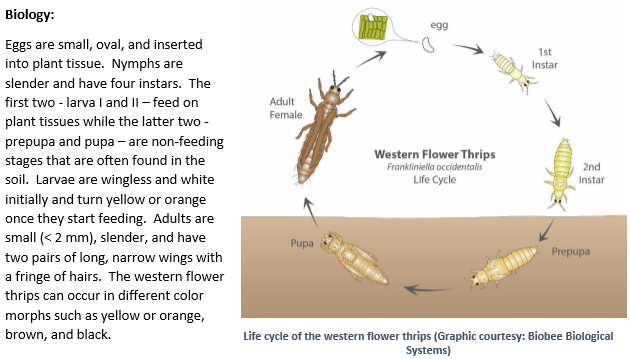
The western flower thrips, Frankliniella occidentalis (Pergande) (Thysanoptera: Thripidae) is one of the major pests of lettuce in California. It has a wide host range including several vegetable, ornamental, and other cultivated or wild plants. Native to North America, the western flower thrips is also known as alfalfa thrips, California thrips, and maize thrips among others. This article provides a general overview of the pest, its biology, damage, and management.
Damage:
The western flower thrips prefers flowers, but also feeds on developing buds, fruits, and foliage. Larvae and adults rupture the leaf surface with their rasping mouthparts and feed on plant juices. Feeding damage results in silvery appearance of the leaf surface, which later turns brown. The presence of dark fecal specs indicates thrips occurrence. In lettuce, the western flower thrips transmits Tomato spotted wilt virus and is the sole vector of Impatiens necrotic spot virus. Only the larval stages acquire these tospoviruses and the adults transmit the viruses to other plants as they spread in the field.
Management: Integrated pest management approach is critical for successful pest management. It involves regular monitoring, exploring the potential of multiple options including cultural and biological solutions, and proper timing and application of various strategies among others. The western flower thrips is one of the pests where insecticide resistance is a common problem. To reduce the risk of resistance development, it is necessary to explore the potential of multiple control options and rotate insecticides with different modes of action. This is essential to suppress pest populations to desired levels and also to maintain control efficacy of existing pesticides.
Cultural control – Remove weed and other hosts that harbor thrips or viruses. Sprinkler irrigation can help reduce thrips populations. Plow down lettuce crop residue to destroy surviving stages. In general, maintaining good plant health with optimal nutrition and irrigation practices helps plants withstand pest damage. Silicate products can improve the structural strength of plant tissues and reduce pest damage and/or populations. Several biostimulants or biological soil amendments can also help activate plant's natural defenses against pest infestations. Consider using them to improve overall plant health and yields, and to protect plants from biotic and abiotic stresses.
Biological control – Predators such lacewings (Chrysopa spp. and Chrysoperla spp.), minute pirate bugs (Orius spp. and Anthocoris spp.), predatory mites (Amblyseius swirski, Ablyseius andersoni, Neoseiulus cucumeris and Stratiolaelaps scimitus), and rove beetles (Dalotia coriaria) attack thrips. Conserve natural enemies with insectary plants and applying safer pesticides, and augment natural populations by releasing commercially reared species.
Microbial control – Entomopathogenic fungi such as Beauveria bassiana and Cordyceps (Isaria) fumosorosea, products based on bacteria such as Burkholderia rinojensis and Chromobacterium subtsugae, and entomopathogenic nematodes such as Heterorhabditis spp. and Steinernema feltiae can be used against one or more life stages. Entomopathogenic nematodes are more effective against pupae in soil because they actively search for and infect their hosts. Entomopathogenic fungi can be used against all life stages.
Botanical control – Azadirachtin alone or in combination with entomopathogenic fungi or insecticides can also be used against multiple life stages. Azadirachtin is an insecticide, antifeedant, and a growth regulator. Similarly, pyrethrins derived from chrysanthemum flowers can be used alone or with other biological or synthetic insecticides. Pyrethrins are nerve poisons. Other botanical insecticides that contain soybean oil, rosemary oil, thymol, and neem oil (which also has a low concentration of azadirachtin) also provide control against thrips through insecticidal, repellency, and antifeedant activities.
Other control options – Insecticidal soaps and mineral oils can be used against different life stages of thrips. Spinosad, a popular insecticide of microbial origin and a mixture of two chemicals spinosyn A and spinosyn D, is very effective against thrips. However, overuse of spinosad can lead to resistance issues in thrips and other insects.
Chemical control – There are several synthetic insecticides that are effective against thrips. It is important to rotate chemicals among different mode of action groups to reduce the risk of insecticide
Chemical control – There are several synthetic insecticides that are effective against thrips. It is important to rotate chemicals among different mode of action groups to reduce the risk of insecticide resistance. The following are some synthetic active ingredients and their mode of actions groups in parenthesis that can be used for thrips control: methomyl (1A), bifenthrin (3A), lambda-cyhalothrin (3A), zeta-cypermethrin (3A), clothianidin (4A), spinetoram (5), and cyantraniliprole (28).
Depending on the level of control needed, combinations of products from different categories can improve control efficacy. For example, a combination of entomopathogenic fungi and nematodes can be applied to the soil for controlling prepupae and pupae. While the soil-dwelling predatory mite S. scimitus and the rove beetle, D. coriaria, can be used against pupal stages, other natural enemies can be used against nymphs and adults. A combination of entomopathogenic fungi and azadirachtin can be applied both to the soil or foliage for controlling different life stages. Similarly, various biological and synthetic insecticides can be applied in combination or rotation to obtain desired control.
The categories presented above are based on the source or nature of the active ingredients and do not indicate their organic or conventional label status. Please check the product labels for their appropriateness for managing thrips in lettuce, for use in organic farms, and guidelines for storage, handling, and field use. Entomopathogenic nematodes, fungi, and other biologicals are compatible with several synthetic agricultural inputs, but verify the label guidelines for specific instructions.
Broad categories of control options for managing western flower thrips
These guidelines can be used for thrips management in multiple crops depending on the label status.
Additional resources:
Dara, S. K. 2019. The new integrated pest management paradigm for the modern age. JIPM 10: 1-9. https://doi.org/10.1093/jipm/pmz010
Dara, S. K. 2021. Biopesticides: categories and use strategies for IPM and IRM. UC ANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=46134
Natwick, E. T., S. V. Joseph, and S. K. Dara. 2017. UC IPM pest management guidelines: lettuce. UC ANR Publication 3450. https://www2.ipm.ucanr.edu/agriculture/lettuce/Western-flower-thrips/
Riley, D. G., S. V. Joseph, R. Srinivasan, and S. Diffie. 2011. Thrips vectors of tospoviruses. JIPM 2: I1I10. https://doi.org/10.1603/IPM10020
Posted by Richard Smith and Joji Muramoto
On March 2, 9 and 16, 2021 we held the sessions of the Organic Soil Fertility Series. The videos of those sessions are now available. The links to each session are listed below along with the topics covered and the presenters.
Part 1: https://youtu.be/WcukYVTeWQg
Estimating Nitrogen Contributions from Cover Crops and Soil Organic Matter and the Role of Soil Microbes in Providing Plant-Available Nitrogen
Presenters: Margaret Lloyd, Daniel Geisseler and Louise Jackson
Part 2:https://youtu.be/Ts1T-Dt2ucw
Estimating Nitrogen Release from Organic Amendments and Irrigation Water
Presenters: Patricia Lazicki, Margaret Lloyd and Michael Cahn
Part 3:https://youtu.be/GoTNhd-eSR0
Putting it all Together: Nitrogen Management in Strawberries and Vegetables and Discussion of New Concepts in Organic Nitrogen Nutrition
Presenters: Joji Muramoto and Richard Smith
Gouda (The Netherlands), 15 April 2021
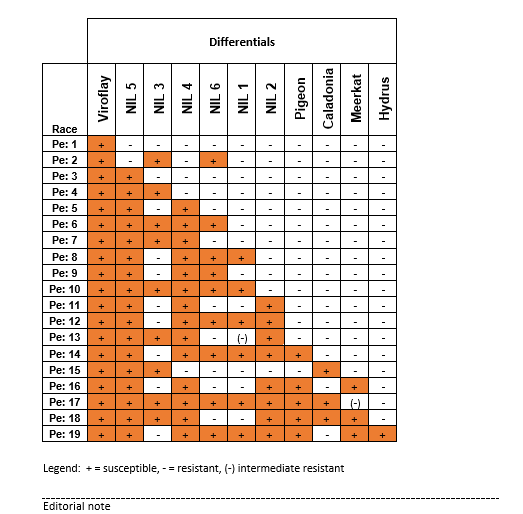
Two new races of the downy mildew pathogen (Peronospora effusa) on spinach have been denominated by the International Working Group on Peronospora in spinach (IWGP) on the basis of a worldwide evaluation of isolates from growers fields and trap nurseries. Isolate SP1924 found in Europe, is denominated as race Pe: 18. Isolate UA202001E, found in the USA, is denominated as race Pe: 19. Both races pose a significant threat to the spinach industry in all parts of the world, and resistance to these new races is important.
Members of the IWGP are using a fixed set of spinach differentials (with different resistances) to define races of downy mildew on spinach by their pattern of virulence on the set. The virulence patterns of all races are published as reference data by the International Seed Federation (ISF); https://www.worldseed.org/our-work/plant-health/differential-hosts/
Race Pe: 18 is able to infect the differentials NIL2, 3, 4, 5, Pigeon, Caladonia, and Meerkat. Pe: 18 has been found in the US in 2015 to 2018, not in 2019 and 2020. And in Europe it has been found more often in the last 3 years. Race Pe: 19 is able to infect the differentials NIL1, 2, 4, 5, 6, Pigeon, Meerkat and Hydrus. Pe: 19 has been reported only from the USA until now.
The IWGP is continuously monitoring the appearance of strains of the pathogen that deviate in virulence from the known races. In this way the IWGP aims to promote a consistent and clear communication between public and private entities, such as the seed industry, growers, scientists, and other interested parties, about all resistance-breaking races that are persistent enough to survive over several years, occur in a wide area, and cause a significant economic impact.
The IWGP is operating internationally and is administered by Plantum located in The Netherlands. The IWGP consists of representatives from spinach seed companies (BASF, Bayer, Bejo, DeSeed, Enza, Pop Vriend, Rijk Zwaan, Sakata, Syngenta, Takii, and Vilmorin) and Naktuinbouw, and is supported by public research at the University of Arkansas. Spinach researchers over the world are invited to join the IWGP initiative and use the common host differential set to identify new isolates. All denominated isolates and seeds of the differential set are available at Naktuinbouw (The Netherlands).
Attached:
Table with disease resistance reactions of spinach downy mildew races on IWGP differentials. Differentials and type isolates are available at Naktuinbouw in The Netherlands.
This table is also available at https://www.worldseed.org/our-work/plant-health/differential-hosts/
For more information on this subject you can contact Jim Correll (jcorrell@uark.edu), Diederik Smilde (d.smilde@naktuinbouw.nl), or the IWGP chairperson Anne Königs (a.konigs@rijkzwaan.nl)
Press Release Pe 18 and 19 - 15 April 2021 Final
- Author: Michael D Cahn
CropManage Hands-On Webinar
Co-sponsored by:
Pajaro Valley Water Management Agency and
Resource Conservation District of Santa Cruz County
When: Tuesday, May 4, 2021, 9:00 AM-12:00 PM
Where: Virtual Meeting via Zoom (link and instructions will be emailed to registrants)
Hosts: Michael Cahn, Advisor, UCCE Monterey and Andre Biscaro, Advisor, UCCE Ventura
Registration fee: $20
A limited number of registration waivers are available in case of financial difficulties. If interested, please email anrprogramsupport@ucanr.edu to discuss before registering. Registration fees do not apply to Pajaro Valley Growers.
Join us for this hands-on webinar to learn to use CropManage to support irrigation and nutrient management decisions and record-keeping. Para el formulario de registro en español, vaya aquí.
What is CropManage?
CropManage is a free online decision support tool for water and nitrogen management. Based on in-depth research and field studies conducted by the University of California, CropManage provides real-time recommendations for efficient irrigation and fertilizer applications—while maintaining or improving overall yield.
Who should participate?
Vegetable and berry growers, ranch managers, other farm staff, and technical service providers are welcome. The webinar is for both new and current CropManage users.
How to prepare?
As this is a participatory webinar, please join via computer or tablet so that you can follow along and participate in the exercises. Two screens will work better so participants can follow the webinar and use CropManage at the same time.
Each participant will need a user account for CropManage. Please set up a free user account at cropmanage.ucanr.edu before the webinar.
Continuing Education
Certified Crop Advisers (CCA) 2.5 hours of CE units have been approved
Contacts for More Information
Logistics & Registration: Rachel Palmer, anrprogramsupport@ucanr.edu, or 530-750-1361
Course Content: Michael Cahn, mdcahn@ucanr.edu; Farm Advisor, UCCE Monterey County
Para obtener más información en español, comuníquese con Sacha Lozano al 831-224-0293
- Author: Michael D Cahn
- Author: David Chambers
Tensiometers are useful for monitoring soil moisture in vegetable and row crops so that plants are not over-watered nor become water stressed. As the name implies, tensiometers measure soil moisture (water) tension, otherwise referred to as matric water potential. Soil water tension is essentially a measure of the force that plants need to exert to suck water from the soil pores into the roots. A high tension means that the soil is dry and a plant needs more energy to extract water from the soil compared to when the soil is moist. Tensiometers function without batteries and wires, although they can be interfaced to dataloggers by adding a pressure transducer. Also, the readings are not affected by soil texture, temperature, and salinity unlike many electronic soil moisture sensors.
A previous article published in this blog described how to build a tensiometer from PVC parts and ceramic cups that can be purchased from a supplier in California. The design results in a dependable and inexpensive tensiometer that can be built by anyone comfortable using a few hand tools. The most challenging step was gluing the cup to the end of the PVC shaft using epoxy cement which can sometimes result in a vacuum leak if the bond is weak. Since publishing this article, we have built and used more than a hundred of these tensiometers and can suggest some modifications to the original design and a few other words of wisdom. For example, we observed that the PVC “T” can crack if the gauge is threaded too tightly or during cold weather conditions which can cause the plastic to contract, so we suggest using a different type of “T” and not to over thread the gauge. We have also collaborated with a supplier of the ceramic cups, SoilMoisture Equipment Corporation, whose engineers have developed a cup that can be bonded to the PVC shaft using standard PVC cement and primer rather than a specialized epoxy (Fig. 1). This improvement greatly reduces the time required to build the tensiometer which is why we call it the “ten-minute tensiometer.” Once you have some experience, you will probably need less than 10 minutes to assemble one of these tensiometers from the parts shown in Fig 2.
Materials needed:
Ceramic cups
Vender: SoilMoisture Equipment Corporation, Santa Barbara CA (805-964-3525, www.soilmoisture.com)
Part Number Y2630C, Dimensions: 0.875 inch OD x 2.75 inch length
Cost: $25 ea. Note that this is a new part that must be special ordered.
Vacuum gauge
Vender: Zoro.com/Grainger.com
Part Number 4FMK3, Description: ¼ inch MNPT 2 inch diameter test vacuum gauge
Weblink: https://www.zoro.com/zoro-select-vacuum-gauge-test-2-in-4fmk3/i/G0040792/?q=4FMK3 Cost: $18.99 ea.
#1 size rubber stopper
Vender: Grainger.com
Part Number 8DWU6, model RST1-S, Description: 24 mm neck, bottom diam. = 14 mm. Top diam. = 20 mm.
Weblink: https://www.grainger.com/product/GRAINGER-APPROVED-Stopper-8DWU6?internalSearchTerm=Stopper%2C+24mm%2C+Black%2C+PK52&suggestConfigId=8&searchBar=true&suggestConfigId=8&searchBar=true&suggestConfigId=8&searchBar=true&suggestConfigId=8&searchBar=true
Cost: $18.55 / 52 pieces
Schedule 40 PVC pipe (½ inch diameter)
Vender: irrigation supply or hardware store
Sand paper (60 or 80 grid)
Vender: hardware store
½-inch PVC “T”
Vender: irrigation supply or hardware store
Description: ½ inch Female slip x ½ inch Female slip T. Note that this T replaces part number 402-072 from Spears Inc., which we found to sometimes split during cold periods. Also, the previous part was often not in stock.
PVC bushing
Vender: irrigation supply or hardware store. Description: ½-inch PVC Male slip x female ¼” thread bushing.
PVC cement (gray) and purple primer (Fig. 3)
Vender: irrigation supply or hardware store. Note that the gray cement provides a stronger bond than the clear product.
Gas pipe thread sealant (white or blue paste type)
Vender: irrigation supply or hardware store
Painters masking tape
Vender: hardware store
Petroleum Jelly (Vasoline)
Vender: pharmacy. A coating of petroleum jelly improves the seal between the rubber stopper and PVC tube.
Rubber gloves
Vender: hardware store, paint store, etc. Description: Nitrile disposable gloves to protect hands from PVC primer and glue.
Tools needed:
- PVC saw
- Miter box
Procedures
- Cut PVC pipe sections in the following lengths
1 foot depth tensiometer: top shaft = 4 inches*, bottom shaft = 17 inches
2 foot depth tensiometer: top shaft = 4inches, bottom shaft = 30 inches
We advise first cutting the bottom shaft about 1-inch longer than indicated above using the miter box or an electric miter saw to assure that the cut at the lower end is at a 90-degree angle (Fig. 4). The ceramic cup will fit crooked on the end of the shaft if the cut deviates from 90- degrees. After assuring that the cup fits well, the top end of the shaft can be cut to the exact length.
*Note that the top shaft can cut shorter than 4-inches so that when the tensiometer is installed in the field it has a lower profile to the ground, thereby reducing chances of being hit by tractor implements.

- Check that the ceramic cup fits into the bottom of the PVC shaft and is aligned straight. Sand the neck of the cup and/or the interior walls of the PVC shaft if the cup cannot be inserted at least half-way into the PVC. Be careful not to over-sand the cup or the fit between the neck and the PVC shaft will be loose and not bond well when glued. The fit between the neck of the cup and the PVC should be very tight such that a lot of force is needed to insert the cup into the bottom of the PVC shaft. If the cup is not in alignment with the PVC shaft, then recut the end with the miter box at a 90-degree angle and recheck the fit.
- Wrap the bottom of the PVC shaft and the top of the ceramic cup with painter's tape to prevent cement from coating the outside of the ceramic cup (Fig. 5).
 Fig. 5 Painter’s tape will protect the outer surface of the ceramic cup from becoming coated with cement and primer.
Fig. 5 Painter’s tape will protect the outer surface of the ceramic cup from becoming coated with cement and primer.- In a well-ventilated location, apply PVC primer to both the interior of the PVC shaft and the outside of the PVC top of the ceramic cup. Then apply gray PVC glue to both surfaces, and push the parts together, and hold in place for about 30 seconds to 1 minute (Figs. 6-9). Tip: slightly twist the parts by about 30-degrees immediately after gluing to assure a good bond before the cement begins to set.
 Fig. 6. Coat the PVC portion of the cup with primer and then PVC gray cement
Fig. 6. Coat the PVC portion of the cup with primer and then PVC gray cement Fig. 7 Coat the inside of the bottom of the shaft with primer and PVC gray cement.
Fig. 7 Coat the inside of the bottom of the shaft with primer and PVC gray cement. Fig. 8 Insert the cup into the shaft.
Fig. 8 Insert the cup into the shaft. Fig. 9 After inserting the cup into the shaft immediately twist the cup about 30-degrees and hold in place for 1 minute while the cement sets. Wipe off any excess cement.
Fig. 9 After inserting the cup into the shaft immediately twist the cup about 30-degrees and hold in place for 1 minute while the cement sets. Wipe off any excess cement. - Next glue the top and bottom shafts and the bushing into the ½ inch, PVC slip “T” using the PVC primer and cement. After inserting each of these parts into the T, slightly twist them and hold in place for about 30 seconds while the cement sets (Figs 10-12). Also cover the non-glued areas with painter's tape to prevent the outside from becoming covered with primer and cement.
 Fig. 10 Add primer to the interior of the “T” and the outside edges of the shafts and bushing. Then coat both surfaces with PVC cement.
Fig. 10 Add primer to the interior of the “T” and the outside edges of the shafts and bushing. Then coat both surfaces with PVC cement. Fig. 11 Hold the parts in place for at least 30 seconds after gluing so that the cement can set.
Fig. 11 Hold the parts in place for at least 30 seconds after gluing so that the cement can set. Fig. 12 The “T” shown with the bushing and upper and lower shafts connected.
Fig. 12 The “T” shown with the bushing and upper and lower shafts connected.- Coat the ¼ inch male threads of the gauge with pipe thread sealant and hand screw on the vacuum gauge. Tip: do not over tighten or the PVC “T” may crack.
- Fill the tensiometer fully with degassed distilled water. The water can be degassed by boiling it and allowing it to cool.
- Coat the lower end of the rubber stopper with a thin film of petroleum jelly and insert into the top end of the tensiometer with a light twist to firmly seat the stopper (A loose stopper is the main cause for vacuum leaks).

Conclusion
So that is it—the tensiometer is ready for testing and installation (Fig. 13). Hopefully, it did not take too long to build and with practice one should be able to assemble these tensiometers in 10 minutes or less. Please visit our previous blog article on how to test and install tensiometers in the field. Please let us know if you have any questions or feed back.