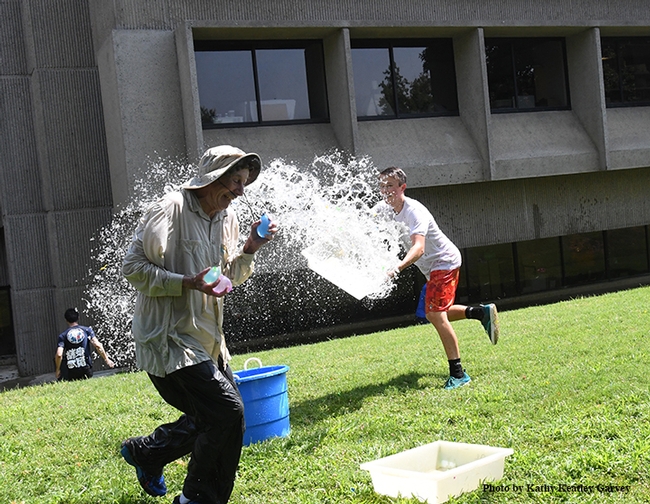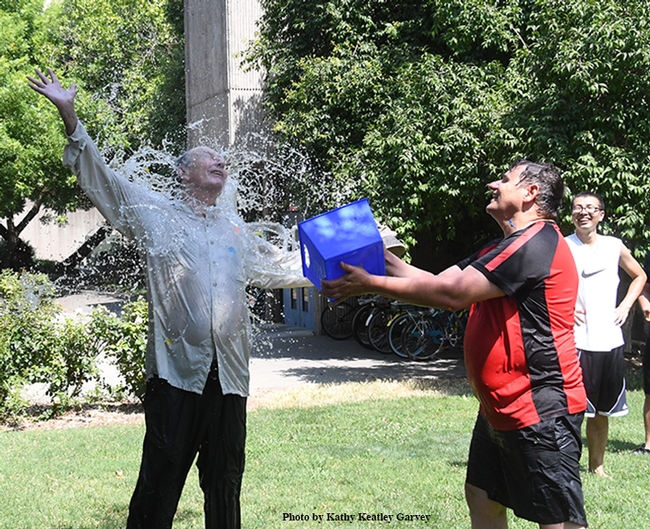- Author: Kathy Keatley Garvey

EicOsis Human Health LLC, the Davis-based pharmaceutical company developing a non-narcotic drug to relieve chronic pain and inflammation, today announced the next step in its ongoing human clinical trials: the initiation of Phase 1b to test the safety of its drug candidate, EC5026.
The ongoing double-blind, placebo-controlled Phase 1b study is designed to investigate the safety and pharmacokinetics of daily doses of EC5026 over seven days.
EC5026, an inhibitor of the soluble hydrolase enzyme (sEH), was discovered and patented at EicOsis based on previous patents and research papers from the laboratory of UC Davis distinguished professor Bruce Hammock during his half-century of research.
“EC5026 plays a crucial role in regulating the metabolism of signaling lipids and responding to inflammation and other stress responses caused by trauma or disease,” said Hammock, who holds a joint appointment with the Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center. “We found sEH to be a target for treating pain, inflammation, cancer and other diseases.”
By inhibiting sEH, EC5026 alleviates pain by preventing the breakdown of natural analgesic and anti-inflammatory fatty acids. Preclinical studies demonstrate no sedation or other adverse behavioral effects and no signs of addiction.
“Many regulatory molecules are controlled as much by degradation as biosynthesis,” Hammock explained. "The epoxy fatty acids rapidly degraded by the sEH control blood pressure, fibrosis,immunity, tissue growth, depression, pain, and inflammation, to name a few processes.”

“The initial results from Cohort 1 appear to replicate the very favorable safety profile we observed in our previous two Phase 1a clinical studies where there were no adverse behavioral, cardiovascular, or neurological effects over five ascending single-dose levels,” said Dr. William Schmidt, EicOsis vice president of clinical development. “The maximum dose level was 5 to 10-fold higher than the anticipated analgesic dose in humans.”
“If Cohort 2 dosing also proceeds without clinically significant adverse effects and a favorable pharmacokinetic profile for once-a-day dosing,” Schmidt continued, “this provides encouragement for initiating our first analgesic efficacy study in the second quarter of 2024.”
EicOsis plans to initiate its first pain patient study in April 2024 “to evaluate safety and analgesic effects in patients with a spinal cord injury who have failed to achieve satisfactory pain relief with existing non-opioid chronic pain medications,” said Cindy McReynolds, EicOsis chief executive officer and former director of research.
“Initiation of the Phase 1b program represents a significant milestone for EicOsis Human Health and demonstrates the success and dedication of our team to make this happen,” said McReynolds, who holds a doctorate in pharmacology and toxicology from UC Davis. “Demonstrating safety in Phase 1b studies will allow us to evaluate efficacy in patients and bring forward safe and effective treatments for several serious diseases. For example, the sEH inhibitors are being considered for use for treatment of cancer in the UC Davis Comprehensive Cancer Center, and neurological disease through the UC Davis Health's designated Parkinson's Foundation Center of Excellence.”

“As a potential anti-inflammatory fatty acid, EC5026 holds promise in addressing neuroinflammation, a key factor in the manifestation and even progression of Parkinson's,” said Zhang, who holds both an M.D. and a PhD. “Ensuring its safety profile in healthy individuals is fundamental for establishing a foundation for further clinical trials. this investigation provides valuable insights into design and overall safety parameters, paving the way for the next stages in advancing this innovative therapeutic approach for Parkinson's disease."

The FDA granted Fast Track status to EC5026 in April 2020 to support the unmet medical need for safe and effective non-opioid analgesics. EicOsis clinical scientist Irene Cortés Puch, who authored the successful application, commented: “Both our commitment and focus at EicOsis Human Health are guided by a genuine concern for the well-being of patients and recognizing the importance of identifying effective pain management alternatives. Therefore, the initiation of this Phase 1b clinical trial is an exciting step in advancing our mission to provide safer and effective treatments.”
Hammock traces his research on chronic pain to his earlier work on how a caterpillar becomes a butterfly; a key enzyme, epoxide hydrolase, degrades a caterpillar's juvenile hormone, resulting in metamorphosis.
A member of the UC Davis faculty since 1980, Hammock founded ;EicOsis in 2011 and served as its chief executive officer until February 2023 when he transitioned to scientific officer and chairman of the board of directors. Highly honored by his peers, he is a member of the National Academy of Inventors and the National Academy of Sciences and received the 2020 Lifetime Achievement Award in Innovation from UC Davis Chancellor Gary May. Hammock has authored or co-authored more than 1400 peer-reviewed publications and holds more than 95 patents in agriculture, environmental science and medicinal chemistry.
EicOsis (pronounced eye-co-sis), derives its name from eicosanoids, “the major backbone of chemical mediators in the arachidonate cascade,” McReynolds said. “It symbolizes the epoxide group in chemistry, which is key to the anti-inflammatory chemical mediators and where the biochemical target called soluble epoxide hydrolase works.”
Approximately 50 million Americans (20 percent of the population) suffer from chronic pain, according to the Centers for Disease Control and Prevention. The annual economic toll is $560 billion, encompassing direct medical expenses, lost productivity, and disability claims.
More information on the Phase 1b multiple-ascending dose clinical study is available at https://clinicaltrials.gov/study/NCT06089837.

- Author: Kathy Keatley Garvey

Hammock, who now holds a joint appointment with the Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center, knows too well what fire can do to victims.
So do other members of the Hammock lab and the Department of Surgery, University of Cincinnati College of Medicine (UC CoM).
They've just published research in the Proceedings of the Natural Academy of Sciences detailing their discovery of a key regulatory mechanism in inflammation that may lead to resolving inflammation in burn patients, as well as sepsis, cancer and COVID patients.
Basically, they discovered a pathway that regulates the immune response after infection or injury, such as burns. Dysregulation of this pathway could differentiate those who are at risk of fatal sepsis or help identify targets to resolve this unregulated inflammation.
“We are very excited about the findings in this paper and the far-reaching impacts it could have on understanding a key regulatory step in the immune response,” said co-lead author and researcher Cindy McReynolds of the Hammock lab and director of research at EicOsis, a Davis-based company founded by Hammock. Hammock, the corresponding author of the publication, has been involved in enzyme research for more than 50 years.

“This dysregulation has fatal consequences in serious diseases such as COVID, cancer, sepsis, burn, where fatality rates can be as high as 40 percent in severe cases,” she said. “An understanding of these pathways can help identify patients at risk of developing serious disease or identify new therapeutic targets for treatment.”

The research, titled "sEH-Derived Metabolites of Linoleic Acid Drive Pathologic Inflammation while Impairing Key Innate Immune Cell Function in Burn Injury,” is co-authored by Debin Wan, formerly of the Hammock lab and now a scientist at Escape Bio, San Francisco; Nalin Singh of the Hammock lab; and three UC CoM researchers: Charles Caldwell, professor and director, Division of Research, Department of Surgery; Dorothy Supp, adjunct professor in the Department of Surgery and a scientific staff member at Shriners Children's Ohio; and Holly Goetzman, principal research assistant in the Caldwell lab.
It's a complicated research project, but a crucial one to help humanity.
And that's what EicOsis is all about, as well. Hammock founded EicOsis in December 2011 to advance novel, safe and effective oral treatments for patients suffering from pain and inflammation. The LLC is developing a new class of oral non-narcotic analgesics based on inhibition of the soluble epoxide hydrolase enzyme. Human clinical trials are underway to test the drug candidate, EC5026, a first-in-class, small molecule that potently inhibits sEH. The sEH inhibitors have already shown to be effective for inflammatory and neuropathic pain in animals, with no apparent adverse or addictive reactions.

- Author: Kathy Keatley Garvey
Of the more than 30 million cases of COVID-19 in the United States, 547,000 people have died. They are not numbers: they represent family, friends, co-workers, colleagues, neighbors and acquaintances who have succumbed to this tragic disease.
And today Dr. Rochelle Walensky, director of the Centers for Disease Control and Prevention (CDC) warns of another surge. Our nation, she says, shows a seven-day average of about 57,000 new COVID-19 cases per day, a 7 percent increase over the last week.
A burning question: Why do some COVID-19 patients recover and some don't?
The laboratory of UC Davis distinguished professor Bruce Hammock, who holds joint appointments with the Department of Entomology and the UC Davis Comprehensive Cancer Center, may have just pinpointed why.

The team of eight UC researchers, primarily from the Hammock lab, found that four compounds in the blood of COVID-19 patients are highly associated with the disease. Their paper, “Plasma Linoleate Diols Are Potential Biomarkers for Severe COVID-19 Infections,” is published as open access in the current edition of Frontiers in Physiology.
ARDS, characterized by fluid build-up in the lungs, is the second leading cause of death in COVID-19 patients, next to viral pneumonia, according to the National Center for Biotechnology Information.
“Different outcomes from COVID-19 infections are both terrifying from a human health perspective and fascinating from a research perspective,” said UC Davis lead author and doctoral candidate Cindy McReynolds of the Hammock lab. “Our data provide an important clue to help determine what impacts the severity of COVID-19 outcomes. Initially, we focused on the immune response and cytokine profile as important drivers in severity, but considering what we now know from our study and others in the field, lipid mediators may be the missing link to answering questions such as why some people are asymptomatic while others die, or why some disease resolves quickly while others suffer from long-haul COVID.”

“The hypothesis advanced in this paper is that because the leukotoxins have been associated with serious illness and death in humans and dogs and the symptoms are those of adult respiratory distress syndrome, these compounds are biomarkers of pulmonary involvement in COVID-19,” Hammock said. “We also think that it is the conversion of leukotoxin to the toxic leukotoxin diol that causes pulmonary and perivascular edema and this could be leading to the respiratory complications.”
“So the leukotoxins and leukotoxin diols,” Hammock said, “are indicators of respiratory problems in COVID-19 patients as plasma biomarkers. They also present a pathway for reducing ARDS in COVID-19 if we could inhibit the soluble epoxide hydrolase, a key regulatory enzyme involved in the metabolism of immune resolving fatty acids.”
The UC Davis scientists used clinical data collected from six patients with laboratory-confirmed SARS-CoV-2 infection and admitted to the UC Davis Medical Center, Sacramento, and 44 healthy samples carefully chosen from the healthy control arm of a recently completed clinical study.
The Hammock lab's 50-year research on soluble epoxide hydrolase (sEH) and its inhibitors led the professor to found and direct EicOsis Human Health, a Davis-based company that is developing a potent soluble epoxide hydrolase inhibitor for pain relief. Epoxy fatty acids control blood pressure, fibrosis, immunity, tissue growth, depression, pain, inflammation and other processes.
But more recently, the Hammock lab has turned its attention to using sEH as a means to resolve inflammation associated with COVID-19 and the fibrosis that can follow.
The paper is the work of Hammock, McReynolds and Jun Yang (corresponding author) of the Department of Entomology and Nematology and EicOsis Human Health; Irene Cortes-Puch of the Department of Entomology and Nematology, EicOsis Human Health, and the Department of Internal Medicine's Division of Pulmonary Critical Care and Sleep Medicine; Resmi Ravindran and Imran Khan of the Department of Pathology and Laboratory Medicine; Bruce G. Hammock of UC Davis Department of Veterinary Medicine, Aquatic Health; and Pei-an Betty Shih of the UC San Diego Department of Psychiatry.
See the news story on the Department of Entomology and Nematology website at https://bit.ly/3lSWbwf

- Author: Kathy Keatley Garvey

And now, the company he founded, EicOsis LLC, to develop a non-opiate drug to relieve inflammatory pain in companion animals and target chronic neuropathic pain in humans and horses, can add “Sacramento Region Innovator of the Year” to its list of accomplishments.
EicOsis won the award in the medical health/biopharmaceutical category of the annual Sacramento Region Innovation Awards Program. The program “recognizes the area's vibrant innovation community—from emerging to established companies—and their breakthrough creations,” according to sponsors Stoel Rives LLP, Moss Adams LLP and the Sacramento Business Journal.
“This project is an illustration of how fundamental science leads to real world applications, in this case addressing severe pain of humans and companion animals,” said Hammock, chief executive officer of EicOsis and a UC Davis faculty member who holds a joint appointment with the Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center. “Our success in translation has been due largely to support from a number of institutes of the National Institutes of Health and a small team of hard-working scientists.”
The ceremony, honoring the winners of the eight categories, took place Nov. 7 in the Crest Theatre, Sacramento. Judges scored the finalists on novelty, market need, economic or social impact and disruption. (See more information on YouTube (the EicOsis award presentation starts at 1:05.)
“It was an honor to be awarded the Sacramento Region Innovator of the Year in Medical Health and BioPharma,” said Cindy McReynolds, senior program manager of EicOsis and a UC Davis doctoral candidate studying pharmacology and toxicology. “The companies represented were inspiring, and it is great to be a part of the innovation going on in the Sacramento region.”
“Chronic pain is an enormous emotional and economic burden for more than 100 million people in the United States alone,” said Hammock, who co-founded EicOsis in December 2011 to alleviate pain in humans and companion animals. “The extreme and poorly treated pain that I observed as a medical officer working in a burn clinic in the Army, is a major driver for me to translate my research to help patients with severe pain.”

Phase 1 human clinical trials to test the drug candidate, EC5026, a first-in-class, small molecule that potently inhibitssEH, will begin Dec. 10 in Texas. The title: "A Single-Center, Double-Blind, Placebo-Controlled, Phase 1a Single Ascending Dose Study to Investigate the Safety, Tolerability, and Pharmacokinetics of Sequential Dose Regiments of Oral EC5026 in Healthy Male and Female Subjects." Eight will participate; six with the drug candidate and two with the placebo. The technology was discovered in the Hammock lab and UC Davis has licensed patents exclusively to EicOsis.
“EC5026 is a key regulatory enzyme involved in the metabolism of membrane fatty acids,” Hammock said. "It's a novel, non-opioid and oral therapy for neuropathic and inflammatory pain. Inhibition of sEH treats pain by stabilizing natural analgesic and anti-inflammatory mediators."
The project is unique in that “there have been very few truly new types of analgesic compounds that have reached the market in the past 50 years,” Hammock said.
“The sEH enzyme is involved in regulating the activity of powerful anti-inflammatory fatty acids called EETs that are present in all cells in humans and animals,” the scientists explained in their awards application. “EETs are anti-inflammatory, analgesic, anti-hypertensive, but they are short lived molecules that are normally eliminated within seconds. By inhibiting sEH, EET levels can be increased by 4x or more and maintained at high anti-inflammatory and analgesic levels for 24 hours or longer.”
“The sEH inhibitors are very potent molecules that are designed for once daily oral dosing. They can also be administered intravenously for acute pain (e.g. equine laminitis),” they wrote. “Preclinical safety studies show that sEH inhibitors are very safe with no visible signs of toxicity at doses more than 100x higher than the therapeutic dose levels. Unlike conventional analgesics, they do not produce sedation or cognitive dysfunction and they have been shown to have no addiction liability, no adverse cardiovascular effects, and no adverse effects on the gastrointestinal tract. They can be safely co-administered with existing analgesic medications.”
Approximately 50 million Americans (20 percent of the population) suffer from chronic pain, according to the Center for Disease Control and Prevention. The annual economic toll is $560 billion, encompassing direct medical expenses, lost productivity, and disability claims. Pain research is now one of the top priorities of the National Institutes of Health (NIH).
EicOsis advancement of EC5026 into clinical trials has been funded as part of the Blueprint Neurotherapeutics Network (BPN) of the NIH Blueprint for Neuroscience Research. The BPN is a collaboration of NIH Institutes and Centers that supports innovative research on the nervous system with the goal of developing new neurotherapeutic drugs.
EicOsis (pronounced eye-cosis), derives its name from eicosanoid, “the major backbone of chemical mediators in the arachidonate cascade,” said McReynolds. “It symbolizes the epoxide group in chemistry, which is key to the anti-inflammatory chemical mediators and where the biochemical target called soluble epoxide hydrolase works.”

- Author: Kathy Keatley Garvey

What do you get?
An epic battle during the 15th annual Bruce Hammock Lab Water Balloon Battle on the Briggs Hall lawn at the University of California, Davis.
Who won? It was not distinguished professor Bruce Hammock, who holds a joint appointment with the UC Davis Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center.
It was undergraduate student Andrew Kisin of the lab of Aldrin Gomes, UC Davis Department of Neurobiology, Physiology and Behavior.
Hammock, clutching three water balloons, didn't stand a chance as Kisin raced toward him with a container full of water. The rest is history. Drenched history.
The annual battle amounts to 15 minutes, or "15 Minutes of Aim." That's how long it takes for the some 40 participants to toss 2,000 water balloons. Currently 28 researchers--from the United Staes, China, France, Ukraine, Lebanon, Japan and Korea--work in the Hammock lab. They include postdoctoral scholars, researchers, graduate students, visiting scholars, visiting graduate students, visiting summer students, short-term visiting scholars and student interns.
Trained as a entomologist, chemist and toxicologist--and who now focuses his research on human health, Bruce Hammock is known for his work on using natural chemical mediators to control inflammation and intractable pain. He co-discovered the soluble epoxide hydrolase, and many of his more than 1100 publications and patents are on the P450 branch of the arachidonate cascade where the soluble epoxide hydrolase (sEH) degrades natural analgesic and anti-inflammatory compounds.
Kisin, a second-year UC Davis student from San Jose, just enrolled at UC Davis a week ago. Majoring in biological sciences, he joined the Gomes lab to study "the effects of drugs such as ibuprofin on organs and may also study how proteasomes contribute to aging."
The annual event is open to all UC Davis personnel and their families. They fill the balloons, toss them and clean up the remnants--and then look forward to next year's battle.
Odds are that you won't find many water warriors like Bruce Hammock, whose credentials rival noted academicians worldwide. A native of Little Rock, Ark., who holds a doctorate from UC Berkeley, Bruce joined the UC Davis faculty in 1980. He is the founding director (1987-present) of the UC Davis NIEHS (National Institute of Environmental Health Sciences) Superfund Research Program and is a founding member (1990-present) of the UC Davis Comprehensive Cancer Center. He has directed the UC Davis NIH/NIEHS Combined Analytical Laboratory for 25 years.
Highly honored by his peers, Hammock is a fellow of the National Academy of Inventors, which honors academic invention and encourages translations of inventions to benefit society. He is a member of the U.S. National Academy of Sciences, a fellow of the Entomological Society of America, and the recipient of the Bernard B. Brodie Award in Drug Metabolism, sponsored by the America Society for Pharmacology and Experimental Therapeutics. He is the first McGiff Memorial Awardee in Lipid Biochemistry. The Eicosanoid Research Foundation recently honored him for work on oxidized lipids.
But on one day in July--for 15 minutes--Bruce Hammock turns from academician to water warrior. And yes, he's excels at that, too! See what happened to his doctoral student Cindy McReynolds in one of the photos below.