- Author: Kirsten Ann Pearsons
“In the coastal areas where spotted wilt is a serious problem ... there is much to be learned concerning the seasonal migrations and local host succession of the thrips”
At first, this looks like a quote from 2020 or 2022 made in reference to the recent outbreaks of Impaciens necrotic spot virus (INSV), a thrips-transmitted disease currently affecting lettuce. But the quote is actually pulled from a UC publication titled Thrips of economic importance in California, authored by Professor Stanley Bailey in 1938.
The lettuce industry here in the Salinas Valley has been hit hard by INSV the past few years, and understanding the biology of the thrips that vectors the virus could be critical for management. But as Bailey noted almost 100 years ago, fully understanding the biology of western flower thrips has been elusive for decades.
In Thrips of economic importance in California, Bailey also noted that thrips in coastal areas tend to spend their summers at higher altitudes, but as native hosts dry up, they can concentrate on nearby crops. I wondered if such a migration could help explain the increase in thrips and INSV pressure the past few falls. With advances in thrips collection methods (i.e., sticky cards) and changes in cropping patterns, I was curious -- could we could observe the migration that Bailey described nearly 100 years ago?

Methods: With help from John Massa (Comgro) and a team from Braga Fresh (Eric Morgan, Katie Chiapuzio, and Jaylen Calabro), I set up a loose transect of 10 sticky card traps at about 4' off the ground (Figure 1). The transect spanned 0.38 miles (610 m) and an elevation change of 325 ft (99 m).
The first traps were deployed on June 5th and the last traps were collected on October 25th. We swapped out cards every two weeks for a total of 10 sets of cards. Some cows used two of the lower traps as scratching posts, so we were limited to 8 traps for most of the trial.
Sticky cards were taken back to the lab to count any thrips that fit the general description of Western flower thrips, Frankliniella occidentalis: less than 2 mm long, overall yellow to brown body color. Some larger, black thrips were occasionally found on traps and were excluded from overall counts.
Hypothesis: Thrips migrate down from the hills in late summer and early fall, increasing the thrips pressure in the valley which could increase the risk of spreading INSV.
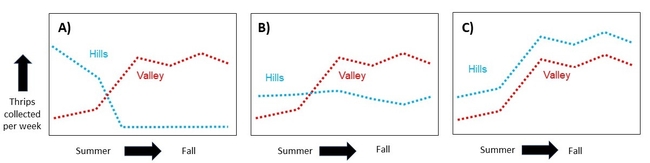
Expected Results: If thrips counts are high in the hills in summer, but drop as populations rise in the valley, then this would be good support for Bailey's note and my hypothesis (Figure 2A). Alternatively, if thrips populations in the hills are consistent across time (Figure 2B), or if their population fluctuations match what is going on in the valley (Figure 2C), then it is unlikely that a mass migration is occurring.
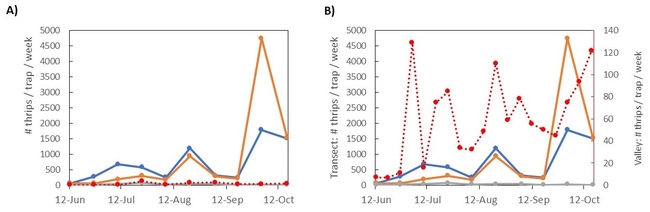
Results: The transect results are summarized in figure 3. In panel A, the average thrips per week is plotted over time, with cards grouped by location (top of the hill, middle of the hill, or towards the bottom). The bottom traps were mostly surrounded by dried grass, while the top and middle traps were generally near chaparral plants that stayed green and flowering throughout the summer and into the fall. You can see an increase in thrips captures from June into July, followed by a dip in early august, and two more peaks in mid-August and early October (following that three-day heatwave). Compared to the valley counts (red line), the number of thrips captured on the hill was much higher, an average of 13 times higher than in the Valley. Adjusting the scale of the Valley-level trap counts (Figure 3B), we can see the Valley traps somewhat followed a similar pattern - thrips populations peaked in early June, had a few weeks of low counts in early August, then peaked again in mid-August and early October. With some variation, adult thrips captures in the hills followed a similar pattern to those captured nearby in the Valley.
Preliminary Conclusions: Contrary to my hypothesis, this small study does not provide evidence that thrips migrate en masse from the hills into the Salinas Valley. The hills maintained some green vegetation and flowers throughout the year, so thrips may not be driven to the Valley like Bailey described. Instead, the hills supported high thrips population throughout the summer and into the fall, which may have acted more like a continuous source of thrips into the valley. This could have interesting effects on INSV epidemiology, depending on whether the host plants in the hills can acquire INSV.
We of course cannot rely on a single transect in one year to conclude that thrips never migrate en mass into the Valley. This year we had an atypical, cool, wet spring that may have changed if or how thrips migrate. Perhaps migration only occurs in years with a drier, warmer spring. We also cannot discount the fact that the thrips we counted may not all be Western flower thrips; the identification characteristics we used (less than 2 mm long, overall yellow to brown body color) are not diagnostic of Western flower thrips. The next steps in this study would be to set up additional transects next year and live collection of thrips off of vegetation. By setting up additional transects (and getting them set up earlier in the season), we could determine if this preliminary transect was an anomaly, or if thrips are not behaving the way that Bailey described in 1938. Live collection of thrips is necessary to determine what proportion of thrips in the hills are Western flower thrips that can vector INSV. Either way, we are one step closer to understanding the seasonal migrations and local host succession of thrips, which could help us in our fight against INSV.
Much thanks to John Massa, Eric Morgan, Katie Chiapuzio, Jaylen Calabro, Jasmine Rodriguez, Luis Ramirez-Espinoza, and Carlos Rodriguez Lopez!
- Author: Kirsten Ann Pearsons
We are getting to the time of year where lettuce production winds down in the Salinas Valley and ramps up in the desert around Yuma, Arizona. Unlike in the Salinas Valley, the desert has not been hit as hard by Impaciens necrotic spot virus (INSV), the virus that is transmitted to lettuce by Western flower thrips (F. occidentalis). When the virus does show up in the desert, the primary infection can often be traced back to INSV-infected thrips that arrived on vegetable transplants from coastal California (Palumbo, 2022)
These finding stirred up concern in the local ag community – could vegetable transplants also be a significant source of new INSV infections in the Salinas Valley? There are instances where recently transplanted fields start showing INSV symptoms soon after planting (Figure 1), but it is challenging to nail down if they were infected before or after transplanting. After gaining some advice from John Palumbo of the University of Arizona (who has been spearheading the ISNV work in the desert), I set out to sample thrips from local transplant nurseries.
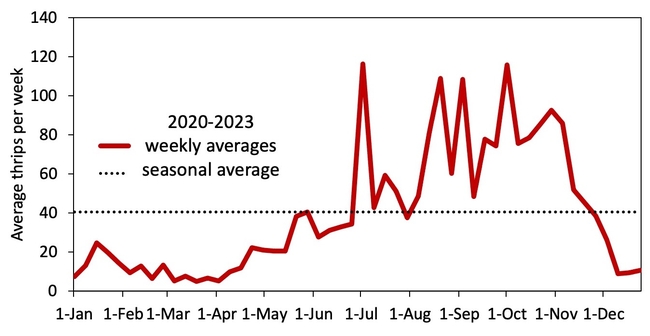
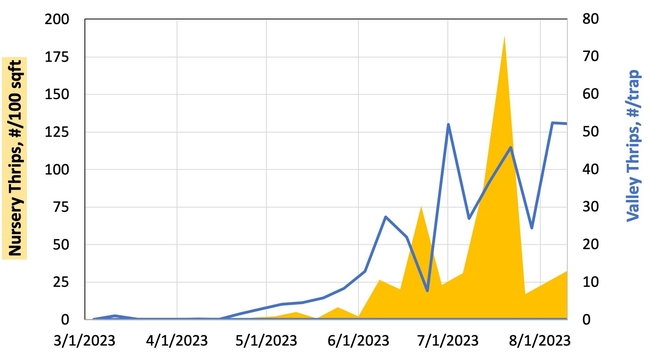
Hypothesis: Vegetable transplants are NOT a major source of INSV-infected thrips in the Salinas Valley. Although this hypothesis may seem contrary to the findings from Yuma, it all comes down to timing of transplant production and background levels of thrips and INSV. As summarized in Figure 2, weekly thrips activity is low in spring and early summer but high in late summer and early spring. [check out an interactive version of these data: Salinas Valley Lettuce Pest Mapping Tool]. This difference in activity somewhat corresponds to when transplants are grown for local use (spring and early summer) versus desert use (late summer and fall). Transplants that are sent to Yuma are grown when thrips populations and INSV pressure has been historically high – putting them at higher risk of carrying INSV+ thrips.

Species confirmation and virus testing: I collaborated with Daniel Hasegawa (USDA-ARS) for species confirmation and virus testing. Daniel and his team used a genetic testing method called multiplex RT-qPCR to determine if the adult and larval thrips were 1) Western flower thrips (F. occidentalis), 2) Onion thrips (Thrips tabaci), or 3) some other thrips species AND if the thrips were carrying 1) INSV or 2) Tomato spotted wilt virus (TSWV, similar to INSV). For small samples (five or fewer thrips), Daniel's team tested each thrips individually. For samples with more than five thrips, they tested half of the thrips as a pooled sample. If this sub-sample tested positive for INSV, a subset of the remaining thrips were tested individually.
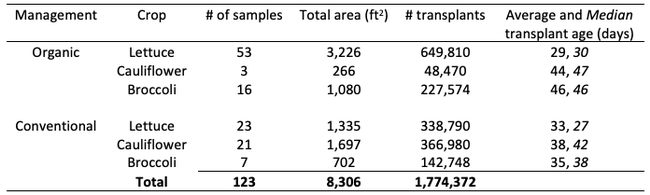
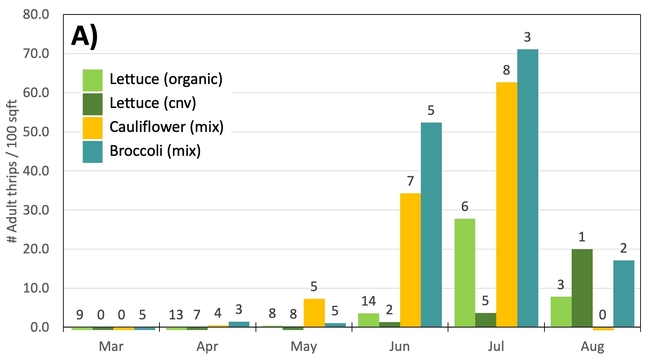
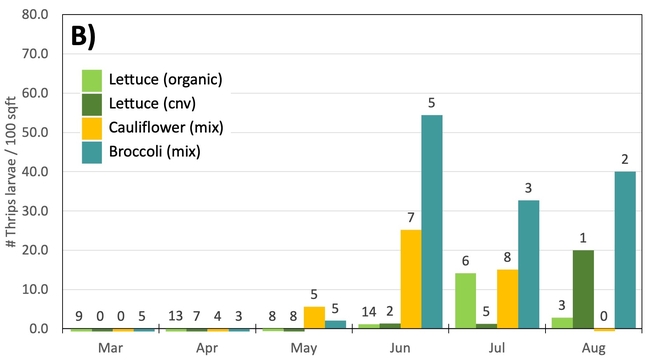
Results: From March through May, we only collected 52 thrips, 50 of which were identified as western flower thrips and none which tested positive for INSV or TSWV. In June, collection shot up almost 10-fold and doubled again in July, somewhat tracking the thrips population increase in the valley (Figure 4). In June and July, thrips densities were consistently higher on brassica transplants compared to lettuce transplants (Figure 5).
Despite collecting over 2,000 individual thrips (1,333 adults and 782 larvae), only two samples, or 1.64% of the tested thrips, tested positive for INSV. The two samples which has positive hits for INSV were collected in June, one off or organic broccoli and one off of conventional cauliflower. None of the thrips collected off of lettuce transplants tested positive for INSV.
Preliminary Conclusions: For the Salinas Valley, the thrips populations on transplants appear to mirror the thrips population in the valley. This is perhaps not too surprising since most transplants are grown on uncovered benches near crop fields. Despite lettuce being a better host for Western flowerthrips (Joseph & Koike, 2021), we consistently collected morethrips frombrassica transplants. We attribute this difference to management practices (e.g., insecticide applications on lettuce transplants) and because brassica transplants tend to stay at nurseries for an additional week.
With this in mind, we may expect that transplanted lettuce fields in the Salinas Valley are more likely to be infected by INSV+ thrips coming in from nearby areas (the prior crop, nearby fields, or weedy areas) rather than thrips from transplant nurseries. Although I would expect to find more INSV+ thrips on vegetable transplants in a year with higher INSV incidence (2023 incidence was considerably lower than prior years), the risk of INSV+ thrips coming in from other sources would increase as well.
For the desert, however, the background risk of INSV is much lower because INSV levels drop off over the summer. In this context, even a handful of INSV+ thrips on transplants pose a proportionally greater risk than in the Salinas Valley. Recognizing this risk to growing regions beyond the Salinas Valley, it is important to continue monitoring INSV levels in transplant nurseries and to work with nurseries to minimize the risk of transporting INSV+ thrips.
References:
- Palumbo, JC, 2022. Thrips and INSV Management in Desert Lettuce. University of Arizona VegIPM Update, Vol 13, No 22, Nov 2, 2022. https://acis.cals.arizona.edu/docs/default-source/agricultural-ipm-documents/vegetable-ipm-updates/2022/thrips-and-insv-management-in-desert-lettuce.pdf?sfvrsn=3088b8b9_2
- Joseph, SV, Koike, ST, 2021. Could Broccoli and Cauliflower Influence the Dispersal Dynamics of Western Flower Thrips (Thysanoptera: Thripidae) to Lettuce in the Salinas Valley of California? Environmental Entomology 50, 995–1005. https://doi.org/10.1093/ee/nvab050
Thanks to Daniel Hasegawa and the entire Hasegawa lab (USDA-ARS, Salinas); John Palumbo (University of Arizona); Kevin Costa, Thomas Costa, and Manuel Aguirre (Headstart Nursery); Francisco Castaneda and Omar Saenz (Growers Transplanting); Lupe Guillen, Maria Alfaro, Alejandro Palma-Carias, and Jim Wilkinson (Dole Fresh Vegetables); Jasmine Rodriguez; Luis Ramirez-Espinoza (CSU-MB); and the California Leafy Greens Research Board.
- Author: Kirsten Ann Pearsons
- Author: Yu-Chen Wang
The 2023 Salinas Fall Pest Management Meeting will be held on the afternoon of Tuesday December 5th, 2023
(postponed from the original date of Nov 2)
Pre-registration is encouraged but not required:
https://surveys.ucanr.edu/survey.cfm?surveynumber=41558
Se recomienda inscripción de antemano:
https://surveys.ucanr.edu/survey.cfm?surveynumber=41560
Traducción simultánea al Español
- Author: Michael Cahn
- Contributor: David Chambers
- Contributor: Noe Cabrera
Growers will need to implement best management practices that reduce nitrate leaching losses on the Central Coast to comply with Agricultural Discharge Order 4.0. The use of drip irrigation has allowed many growers to be efficient with both water and nitrogen fertilizer. Fertigating through the drip system allows for spoon feeding nitrogen in amounts matching the pattern of crop N uptake, and to place fertilizer in the root zone. Tools like the soil nitrate quick test and the online irrigation and nutrient management platform, CropManage, can help farm managers accurately determine the right amount of fertilizer to apply to satisfy crop N requirements without jeopardizing production.
Once the right amount of fertilizer to apply has been determined, it is important that irrigators have the tools that they need to accurately inject the correct volume into the drip system. Fertigation trailers usually consist of a nurse tank that can hold a maximum volume of 500 to 1000 gallons of fertilizer and are equipped with a small gas or electric pump used to inject liquid fertilizer into the drip system. Often irrigators rely on markings on the side of the nurse tank to determine the volume of fertilizer that they are injecting. These markings are usually not accurately calibrated nor have fine enough graduations to precisely measure out fertilizer volume. Furthermore, tank markings can be hard to read, especially if the trailer is not level.
A flowmeter could increase the precision of metering fertilizer into a nurse tank or for measuring the volume of fertilizer injected into the drip system. Using a flowmeter for metering fertilizer would also facilitate tracking the volume of fertilizer applied to each crop by either noting the meter readings or by interfacing the flowmeter to a datalogger that can record the application volumes.
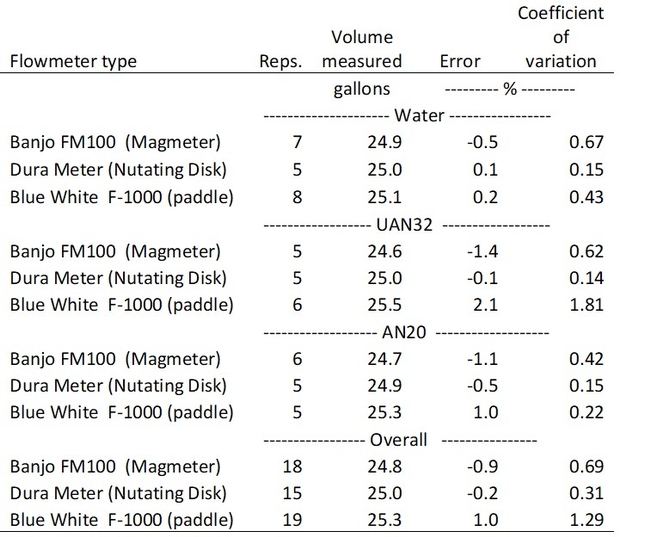
We evaluated the accuracy of three models of flowmeters designed for metering liquid fertilizer: 1. Banjo FM100 meter, 2. Dura-meter, and 3. Blue White F-1000 (Fig. 1). Each model relies on a different mechanism to monitor fertilizer volume. The Banjo meter measures flow using a magnetic sensor, while the Dura-meter uses a nutating disk, and the Blue white meter uses a small propeller. The accuracy of the flowmeters was tested using 25 gallons of either water, ammonium nitrate (20% N), or urea-ammonium nitrate (32% N). A testing manifold was set up in the UCCE Monterey greenhouse that pumped a calibrated volume of each fluid through the flowmeters using an electric diaphragm pump. Five or more test runs were made for each meter and fluid. The average volume measured and standard deviation from the mean volume was calculated.
All three models of flowmeters accurately measured water and fertilizer volumes (Table 1). Measurement errors were generally less than ±2% of the true volume. The Dura-meter which uses a nutating disk to measure volume was the most accurate flowmeter of the three models and had an overall average absolute error of -0.2 gallons per 25 gallons measured, and a coefficient of variation of ±0.3%. The Blue White meter, which uses a paddle wheel to measure volume, was least accurate and had an overall absolute error of 1 gallon per 25 gallons measured and a coefficient of variation of ±1.3%. The type of liquid metered affected the accuracy of the Banjo and Blue White meters more than the Dura-meter.
Table 1. Accuracy of flowmeter measurements of water and two types of liquid fertilizer (AN20 and UAN32).
Although the Dura-meter was most accurate of the three flowmeters, it did require an initial calibration before testing began. The other meters could not be manually calibrated. The nutating disk mechanism directly measures volume of a liquid which may explain why the Dura-meter was not affected by the density of the liquid tested. Both the paddle wheel and the magnetic sensor mechanisms used in the Blue-White and Banjo meters indirectly estimate flow rate. Another advantage of the Dura-meter was that it was the cheapest of the three meters when the tests were conducted. Another version of the Dura-meter can be used to turn off an injection pump when a specified volume of fertilizer has been injected. This version is available as part the auto batch system (Dura-ABS™). The Banjo meter is also available in a model (MFM100) the can output an electrical pulse proportional to flow rate so that volume of fertilizer injected can be recorded on a datalogger.
Conclusions
Three commercially available flowmeters were demonstrated to accurately measure fertilizer. Either of these meters could help irrigators more precisely apply the intended volume of fertilizer to a crop as well as verify and maintain records of the fertilizer volumes used to grow each crop. Depending on the practices of the growing operation it may be more efficient to install the meters on either the nurse tank trailer or the main fertilizer tank. If the nurse tank is used for injecting fertilizer at several fields during the day, then installing the meter on the trailer would be logical, but if the nurse tank is only filled for a single field at a time, the flowmeter could be installed on the main fertilizer tank.
- Author: Michael Cahn
- Contributor: David Chambers
- Contributor: Tom Lockhart
- Contributor: Noe Cabrera
Minimizing suspended sediments in irrigation runoff is desirable for several reasons. For growers reusing tailwater for watering their crops, they must assure that the water has minimal food safety risks by testing it for generic E coli and/or treating it with chlorine. The concentration of free (or reactive) chlorine is reduced when tailwater contains a high concentration of suspended sediments. Treating a large volume of tailwater with chlorine can be a significant expense over a season so it is important to be able to remove as much of the suspended sediments as possible before treatment.
A second reason is that water quality regulations under Agriculture discharge Order 4.0 requires tailwater discharged into public water ways to not be toxic to aquatic organisms. Pesticides that strongly bind to soil, such as pyrethroids, are carried on the suspended sediments in runoff which can cause toxicity to aquatic organisms that live in creeks and rivers downstream from farms. Also, particulate forms of N and P which bind with the suspended sediments pose a water quality risk to receiving waterbodies such as the sloughs and wetlands along the coast. Both nutrients can spur algal blooms which reduces dissolved oxygen available to fish and other aquatic organisms.
In a previous article we discussed a new approach to using Polyacrylamide (PAM), an inexpensive polymer molecule for reducing soil erosion, to treat sprinkler water. This practice uses a specialized applicator (Fig. 1) to condition water flowing from a well with PAM. An advantage of this method is that the cartridges in the applicator release a small amount of PAM (1 to 2 ppm) into the irrigation water, which flocculates soil particles that could potentially become suspended and transported in runoff. Field tests using a prototype version of this applicator resulted in about 90% less suspended sediment in the tailwater when treated with PAM compared to untreated irrigation water.

Auger ditch applicator
A second approach we developed for reducing suspended sediment in runoff is to use a smart applicator that can automatically apply dry PAM to the runoff water flowing in farm ditches. This type of applicator is suspended on a platform above a ditch and uses a hopper filled with dry PAM and an auger system controlled by an electric motor and small computer to drop PAM down a tube into the flowing runoff (Fig. 2). A weir and float mechanism located upstream are used to monitor the flow rate of the runoff so that the computer can adjust the frequency that PAM is applied. A video at this link demonstrates how the auger applicator operates.

Field testing of the ditch applicator
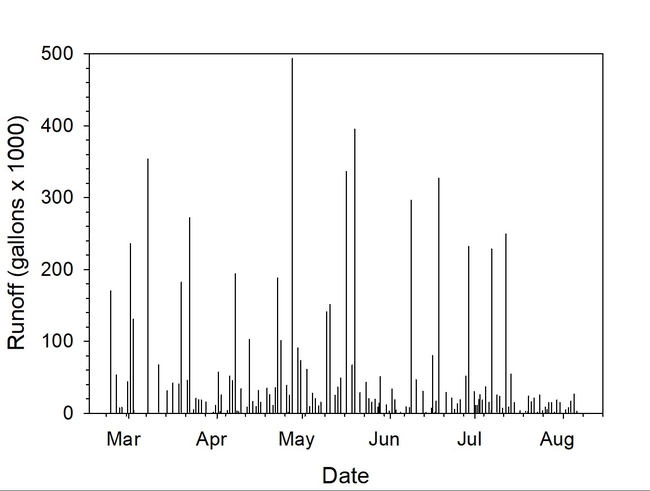
A yearlong study at a commercial farm showed that the ditch applicator was effective in removing 98% of the suspended sediments transported in runoff (Table 1, Fig. 3). Based on the total runoff measured in a single drainage ditch during the 2022 season (21.5 acre-feet), an estimated 106 tons of sediment were removed (Fig. 4).
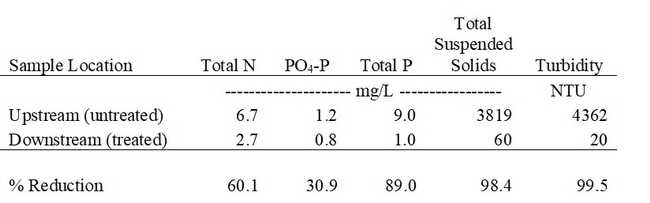
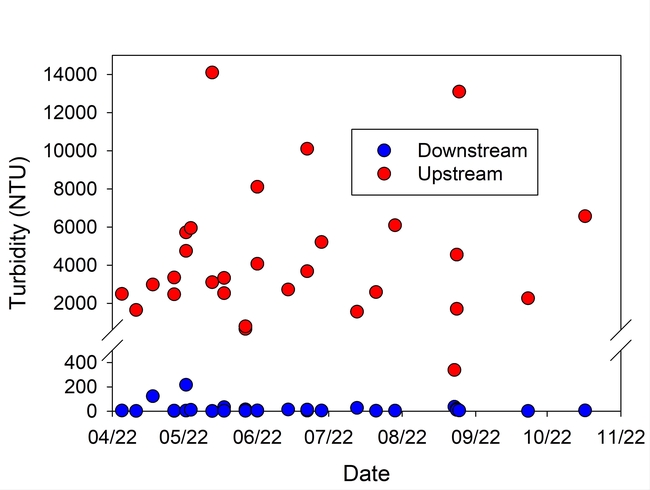
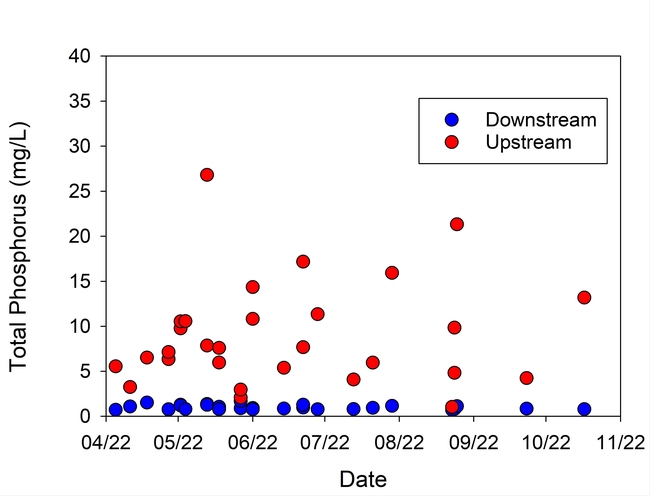
Turbidity in the runoff was reduced by more than 99%, and Total P and N were reduced on average by 89% and 60%, respectively, during the season (Table 1, Figs. 5 and 6). These reductions in nutrient load, suspended sediment, and turbidity could greatly improve water quality in water bodies downstream from farms that discharge irrigation runoff.
Table 1. Average concentration of N, P, and sediments carried in irrigation runoff before (upstream) and after (downstream) treatment with the PAM ditch applicator (April – October 2022). Average of 32 paired grab samples from 3 farm ditches. Downstream locations varied from 300 to 500 ft downstream from the PAM applicators.
Ditch applicator vs well applicator
Although more effective at reducing suspended sediment in runoff than the well applicator, the ditch applicator required more maintenance. PAM needed to be added to the hopper once or twice per week during the irrigation season, and sediment that settled in the ditches had to be cleaned out periodically using a backhoe. Also, removed sediment had to be spread back in the fields. The well applicator only required periodic refilling of the cartridges with PAM, and minimizes the amount of sediment that settles out in the drainage ditches.
PAM effects on chlorine requirement
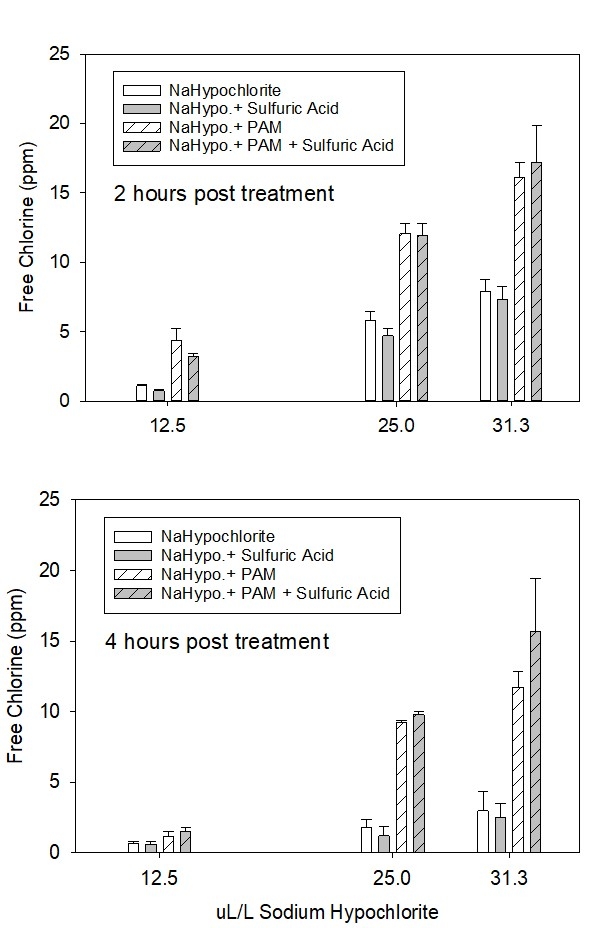
To evaluate the effect of PAM on the quantity of chlorine needed to treat runoff, we performed a laboratory assay on samples of sprinkler runoff collected upstream and downstream of one of the ditch applicators. The turbidity of the upstream (untreated) and downstream samples (PAM treated) was 2276 and 9.5 NTU, respectively. The electrical conductivity of the runoff samples was 1.35 dS/m and the pH was 8.4 before adding chlorine. The main factors evaluated in the assay were sodium hypochlorite concentration and acidification with 10% sulfuric acid. Presumably, acidifying the runoff to a pH of 6.5 should increase the concentration of the more reactive form of chlorine, hypochlorous acid which is more effective as a microbial disinfectant. Residual free chlorine concentration of the treatments was evaluated 2 and 4 hours after adding 12.5% sodium hypochlorite at concentrations ranging 12.5 to 31.3 ul per liter of runoff (100 to 250 ul of 12.5% NaOCl per L of water).
The laboratory assay showed that reducing suspended sediment concentration using PAM increased the efficacy of chlorine treatment of runoff. The free chlorine concentration for PAM treated runoff was more than twice the concentration measured in the untreated runoff for all sodium hypochlorite concentrations evaluated after 2 hours and more than three times the concentration after 4 hours (Fig. 7). Free chlorine concentration in the PAM treated runoff was more than 2.5 ppm two hours after treatment at the lowest concentration of chlorine evaluated (12.5 ul/L) but was less than 0.5 ppm in the untreated runoff. To attain similar chlorine efficacy as PAM treated runoff, untreated runoff would require twice as much sodium hypochlorite (25 ul/L). These chlorine requirements would correspond to 26 and52 gallons of 12.5% sodium hypochlorite to treat and acre-foot of runoff with and without a PAM pretreatment, respectively.
Acidification of the runoff to a pH of 6.5 with sulfuric acid increased the free chlorine concentration in the PAM treated runoff at the highest concentration of sodium hypochlorite (31.3 ul/L) after 4 hours. Acidification did not have a significant effect on free chlorine concentration for the other treatments.
Summary
Both versions of the dry PAM applicators (well and ditch) show promise for greatly reducing soil erosion, as well as helping improve water quality and the efficacy of chlorine for treating tail water reused for irrigation. By considerably reducing the concentration of suspended sediment in irrigation runoff, chlorine can be more effective as a disinfection agent, and better control E. coli and other microbial pathogens that could potentially cause public health risks.
Acknowledgments: We greatly appreciate assistance in fabricating the prototype PAM applicators from RayFab. This project was funded by the California Leafy Green Research Board.
Further reading