- Author: Kathy Keatley Garvey

The question is crucial “because it is the foundation of essentially all biological questions,” says spider systematics researcher Lacie Newton, a doctoral student in the Jason Bond laboratory, UC Davis Department of Entomology and Nematology, and the lead author of newly published research that explores that question.
“For example,” Newton says, “making successful conservation efforts depends on knowing how to identify the threatened/endangered species from other closely related species that are not threatened.”
Her research on folding-door spiders or the Antrodiaetus unicolor species complex led to a journal article published in Molecular Ecology: “Integrative Species Delimitation Reveals Cryptic Diversity in the Southern Appalachian Antrodiaetus unicolor (Araneae: Antrodiaetidae) Species Complex.” UC Davis co-authors are Professor Bond, who is the Evert and Marion Schlinger Endowed Chair in Insect Systematics, and project scientist James Starrett of the Bond lab.
Folding-door spiders are so named because they close the entrances to their silk-lined burrows by pulling in the rim. They are often described as having stocky brown bodies, thick legs and large fangs.
The five-member research team, also including Professor Brent Hendrixson of Millsaps College, Jackson, Miss., and postdoctoral fellow Shahan Derkarabetian of Harvard University, used an integrative approach with several lines of evidence (morphological, behavioral, molecular, and ecological data) to form a consensus “about where we should draw the lines between species in this complex,” Newton said.
They targeted the Antrodiaetus unicolor species complex, which Newton said, are “great organisms for exploring species boundaries because even though these spiders do not have any obvious visual differences to tell them apart--with the exception of the smaller and lighter brown A. microunicolor-- there are significant genetic differences between certain populations, that is potential 'cryptic' species.”

Said Professor Bond of the journal article: “I think its significance lies in the innovative and multipronged approach (integrative) she employed to evaluating species boundaries. The study emphasizes the importance of using both genomic scale and ecological data rather than relying on traditional morphological features alone to delimit species. Understanding species boundaries is an imperative for cataloging and describing the planet's rapidly disappearing biodiversity.”
Newton won a second-place award for her oral presentation on species delimitation at the 2019 American Arachnological Society (AAS), held at Washington and Lee University, Lexington, Va. Her abstract: “Although species delimitation can be highly contentious, the development of reliable methods to accurately ascertain species boundaries is a fundamental and necessary step in cataloguing and describing Earth's quickly disappearing biodiversity. Species delimitation in spider taxa has historically been based on morphological characters; however, certain mygalomorphs are morphologically indistinguishable from each other yet have considerable molecular divergence." She is active in both AAS and the Society of Systematic Biologists.
First-Generation College Student
Newton, a first-generation college student, is a fifth-year doctoral program student whose research interests include systematics, Araneae, mygalomorph spiders, speciation pattern and process, phylogeography, molecular phylogenetics, and character evolution. She is the recipient of a year-long UC Davis Graduate Research Mentorship Fellowship that supports promising doctoral students that meet diversity criteria.
Born and raised in Eupora, Miss.-- “a very small town with less than 2000 people”--Lacie recalls a childhood that included “a significant amount of time outdoors with my family surrounded by the rich flora and fauna of the Coastal Plain Floristic Province.”
“This experience,” she related, “fostered my interest in biodiversity and later guided me to take additional science classes to learn more about the complexities of the living world.”

What sparked her interest in spiders? “I actually used to be terrified of spiders,” Lacie acknowledged. “It wasn't until fall semester of my sophomore year when I took a zoology course that I began to appreciate not only the vast amount of diversity within spiders but also how amazing they are as a group, such as the tensile strength of spider silk being comparable to steel, spider venoms playing a role in potential medical applications, and a myriad of feeding strategies, etc..”
“As my professor Dr. Brent Hendrixson shared his research interests (systematics of mygalomorph spiders and scorpions) and passion for scientific outreach, I evolved from a guarded student to a fascinated one. Additional summer field courses focused on the biology, evolution, and ecology of arachnids completely changed my career trajectory from becoming a medical doctor to an evolutionary biology professor with research emphasizing evolutionary processes of arachnid study systems, specifically mygalomorph spiders like Antrodiaetus.”
Career as Evolutionary Biologist
The UC Davis doctoral student plans a career as an evolutionary biologist, exploring the evolutionary history of mygalomorph spiders. “My ultimate career goal is to become a biology professor where I can perform research and teach in a vibrant academic setting,” Newton said. “As a professor, my aims are to become an expert in my desired field of evolutionary biology, continue to be involved in the scientific community through collaborations with researchers, and become an advocate for vision and change in science education. “
“To clarify,” she added, “I want to study the evolutionary history of arachnids by using emerging technologies/methods and bioinformatics tools. I also plan to participate in the scientific community by publishing articles in respected journals, attending and presenting at conferences, and collaborating with various researchers. Lastly, I plan to take part in teaching and outreach opportunities to convey my enthusiasm for science to others. I feel outreach is especially important to get children enthusiastic about science and to demystify science for the public.”
Newton aims to become a faculty mentor “who can positively impact students--the way my own undergraduate mentor Dr. Hendrixson affected my life--by using my position as a professor to extend opportunities to mentor high school students, undergraduate students, and graduate students, especially from underrepresented groups such as women and members of the LGBTQ community. Specifically, I want to mentor students about career options available as well as offer my own point-of-view about pursuing a career in a STEM field.”
At UC Davis, Newton served as a teaching assistant for the “Introduction to Biology: Biodiversity and the Tree of Life” course. She is mentor to undergraduate students in the Mentoring Program, Equity in Science, Technology, Engineering, Math, and Entrepreneurship (ESTEME) organization, a graduate student organization dedicated to improving equity and inclusion in STEM fields, entrepreneurship, and leadership positions. She also volunteers on the admissions committee for GOALS, the Girls' Outdoor Adventure in Leadership and Science, a summer science program for high school students to learn science hands-on while backpacking through the wilderness.
In addition, Newton volunteers at the annual UC Davis Biodiversity Museum Day and at the Bohart Museum of Entomology open houses, including one featuring “Eight-Legged Wonders (Spiders).”
The "eight-legged wonders," as she said, fascinate her. It's "not only the vast amount of diversity within spiders but also how amazing they are as a group."

- Author: Kathy Keatley Garvey

As environmental artists, bees are "responsible for the brilliantly colored flowers in our landscapes," and as environmental engineers, they engineer “the niches of multitudes of plants, animals and microbes.”
Page, with UC Davis roots and Arizona State University wings, has just authored a 256-page book, “The Art of the Bee: Shaping the Environment from Landscapes to Societies” (Oxford University Press), to be published Aug. 6.
“It's a long time in the making,” said Page, who received his doctorate in entomology at UC Davis and served as a professor and chair of the Department of Entomology (now Entomology and Nematology) before heading to Arizona State University (ASU), where he advanced to school director, college dean and university provost.
“Twenty-five years ago, my friend and mentor Harry Laidlaw (for whom the UC Davis bee facility is named) wanted to write a honey bee biology textbook,” Page recalled. When they finished the outline, “it looked very much like the excellent book by Mark Winston The Biology of the Honey Bee, published in 1987 by Harvard University Press. I decided we didn't need another one, and we still don't.”
The book differs in that it's a collection of “sparkling essays” that “read like mystery stories,” said Rudiger Wehner, professor and director emeritus of the Institute of Zoology, University of Zürich. “With these lucidly written stories, Page takes us on a delightful journey through the many biological traits that on the whole constitute the honeybees' social contract.”
“But don't be fooled by the amiable and personal style—the book is comprehensive—from colony collapse disorder to colony-level evolution—and chock full of the latest results, presented with clarity and depth, leavened with razor-sharp insights into social evolution,” noted Gene Robinson, director, Carl R. Woese Institute for Genomic Biology and Department of Entomology, University of Illinois at Urbana-Champaign.
Page said his book is geared toward “the person who has a basic knowledge of biology and a fascination with bees, perhaps an educated hobby beekeeper--there are a lot of them--or an undergraduate or graduate student with an interest.”

In addition to chapters on environmental artists and environmental engineering, Page includes chapters on social contracts, superorganisms, reproductive competitions, and concludes with “The song of the queen.”
In the epilogue, Page ponders the complexity of individual bees and their colonies, comparing them to humans. “Members of complex societies live close together in closed nests, shared home sites, villages, etc., or in closely connected nomadic tribes. As groups, they typically have a set of tacit rules by which they live that involves working for the good of the group, systems of group and resource defense, internal mechanisms of policing cheaters that don't cooperate and live by the rules, a division of labor often associated with group defense and gathering and sharing resources, and usually asymmetries and rules associated with reproduction. These same general characteristics seem to apply broadly across eusocial insects (aphids, termites, bees, ants, and wasps), eusocial rodents (naked mole rats), higher apes, and humans. Why? The similarities are inescapable due to the nature of social contracts; they must have specific elements to protect the power and will of individuals, whether citizens of the United States of America or workers in a honey bee colony. The contract binds individuals to a society, but the specific social organization evolves by reverse engineering. Natural selection acts on the whole colony; social structure evolves to fit the needs of the group within a given environment. “

Page points out that “Anthropocentric thinking can obscure the way we view nature and lead to false conclusions. Look at Aristotle and honey bee division of labor: For more than 2,000 years it was thought that the bees that work in the nest were postpubescent old men because they're hairy! In fact, the older bees forage and aren't hairy because the hairs break off as they age. I now see my work in a new light; we aren't so different, bees and humans. The elements of our social structures, and how they come about, have many similarities.”
Page is known for his research on honey bee behavior and population genetics, particularly the evolution of complex social behavior. One of his most salient contributions to science was to construct the first genomic map of the honey bee, which sparked a variety of pioneering contributions not only to insect biology but to genetics at large.
At UC Davis, he maintained a honey bee-breeding program for 24 years, from 1989 to 2015, managed by bee breeder-geneticist Kim Fondrk at the Harry H. Laidlaw Jr. Honey Bee Research Facility. They discovered a link between social behavior and maternal traits in bees.
UC Davis named him the 2019 distinguished emeritus professor. Nominator Steve Nadler, professor and chair of the UC Davis Department of Entomology and Nematology, praised Page as “a pioneer researcher in the field of behavioral genetics, an internationally recognized scholar, a highly respected author, a talented and innovative administrator, and a skilled teacher responsible for mentoring many of today's top bee scientists…he is arguably the most influential honey bee biologist of the past 30 years.”
Page has authored more than 250 research papers, including five books. Among them “The Spirit of the Hive: The Mechanisms of Social Evolution” (Harvard University Press, 2013) and “Queen Rearing and Bee Breeding,” with Harry H. Laidlaw (Wicwas Press, 1997). He is a highly cited author on such topics as Africanized bees, genetics and evolution of social organization, sex determination, and division of labor in insect societies.
Page, who received his doctorate in entomology from UC Davis in 1980, joined the UC Davis faculty in 1989 and left as emeritus chair of the Department of Entomology in 2004 when ASU recruited him for what would become a series of top-level administrative roles. He advanced from director of the School of Life Sciences to dean of Life Sciences; vice provost and dean of the College of Liberal Arts and Sciences; and university provost. Today he holds the titles of provost emeritus of ASU and Regents professor emeritus, as well as UC Davis department chair emeritus, professor emeritus, and UC Davis distinguished emeritus professor.
Page is an elected member of the American Academy of Arts and Sciences, the Brazilian Academy of Science, Leopoldina (the German National Academic of Science), and the California Academy of Science. He is a recipient of the Alexander von Humboldt Senior Scientist Award (Humboldt Prize, 1995), the Carl Friedrich von Siemens Fellowship (2013), James W. Creasman Award of Excellence at ASU (2018).


- Author: Kathy Keatley Garvey
(Editor's Note: Geoffrey Attardo, assistant professor, UC Davis Department of Entomology and Nematology, published this piece July 29, 2020 on The Conversation website. This article is republished from The Conversation under a Creative Commons license. Read the original article.)
Bloodthirsty tsetse flies nurse their young, one live birth at a time – understanding this unusual strategy could help fight the disease they spread

By Geoff Attardo, University of California, Davis
Tsetse flies are bloodthirsty. Natives of sub-Saharan Africa, tsetse flies can transmit the microbe Trypanosoma when they take a blood meal. That's the protozoan that causes African sleeping sickness in people; without treatment, it's fatal, and millions of people are at risk due to the bite of a tsetse fly.
My entomology research focuses on insects that feed on the blood of people and animals. From a human health standpoint, understanding what makes all these bugs tick is key to developing ways to control them and prevent transmission of the diseases they carry, such as malaria, dengue, Lyme disease, West Nile virus and many others.
Tsetse flies stand out from their blood-feeding cousins the mosquitoes and ticks because of their unique reproductive biology. They give birth to live young and, even more unusual, the mother lactates and provides milk for her offspring. Here's how it all works – and why their unusual reproduction strategy might be a key to controlling tsetse flies and the parasite they carry once and for all.
From egg to larva
Scientists know of other flies that hold onto their eggs in their reproductive tract until they hatch into young larvae, with each brood consisting of dozens of offspring. The mother then tries to find a suitable source of nutrition in the environment, deposits the larvae and leaves them to survive on their own. The mother does not provide any nutrition for her young.
That's the standard fly way of life. Tsetse flies take a different approach.

Female tsetse flies develop just one single egg at a time. When the egg is complete, the mother moves it from her ovaries into her uterus in a process called ovulation. Once in the uterus, the egg is fertilized with sperm the female has stored in an organ called the spermatheca. While females can mate multiple times, they obtain all the sperm they need for their lifetime from a male fly during a single mating event.
After fertilization, the female keeps the egg in her uterus for five days while an embryo develops within the egg. When the embryo is ready, the egg hatches in the uterus of the female and the tsetse fly larva begins its life living inside its mother's uterus.
Milk meals for baby
Here's where tsetse flies dramatically diverge from most other insects.
Attached to the mother's uterus is a specialized gland that makes a milk-like substance. The organ is called the milk gland, and it produces a rich mixture of fats and particular proteins that provide the larva with all the nutrition it needs to develop into an adult.
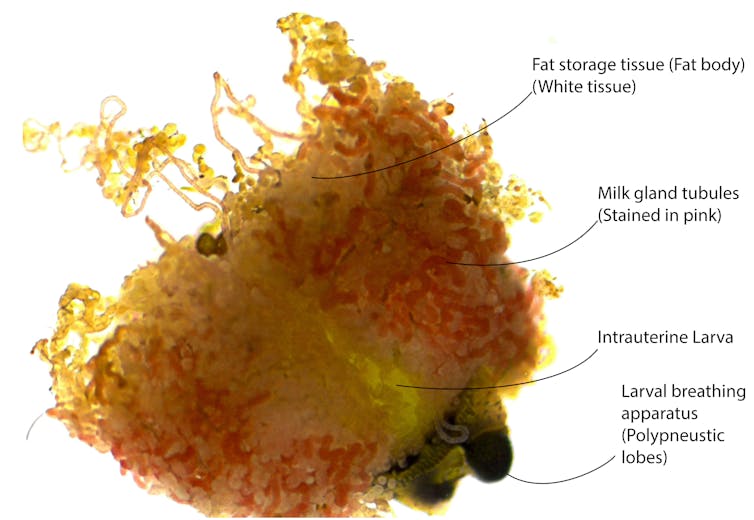
Amazingly, many tsetse milk proteins are very similar in function to those found in the milk produced by mammals.
Just like in mammals, the milk also transfers beneficial bacteria from the mother to the offspring. These bacteria are essential for tsetse flies, and without them adult female flies are unable to reproduce.
After five or six days of developing and feeding on milk, the larva is fully grown and ready to enter the world. The mother finds a safe spot and gives birth. The larva immediately burrows underground to avoid predators and parasites.
Once buried, the outer surface of the larva's skin hardens and turns black, forming a protective shell. This is called the pupal stage and it lasts for around three weeks. During this time, the pupa transforms into an adult fly.

It then emerges from the pupa, climbs out of the ground, and begins its life as an adult tsetse fly looking for hosts to blood-feed on and other tsetse flies to mate with.
Why live birth?
Why would an insect evolve this slow and resource-intensive way to reproduce?
One idea is that this method provides a defensive advantage relative to free-living larvae against parasites and predation. Larvae on their own have few (if any) ways to defend against these threats. But keeping larvae in the mother's uterus provides shelter and a guaranteed food source. While this strategy is much slower, scientists think the extra maternal care results in higher larval survival rates. It's a matter of quality over quantity.
A result of this reproductive strategy is that tsetse fly populations are small and slow to recover from control efforts, relative to more prolific insects like mosquitoes.
My colleagues and I hope that we can parlay our understanding of the molecular processes that regulate tsetses' milk production and mating behavior into new environmentally friendly, cost-effective and tsetse-specific control strategies for these insects.
The sleeping sickness tsetse flies spread is a potential issue for millions of people in 36 sub-Saharan countries, though the number of annual cases has decreased drastically thanks to major control efforts – including trapping flies, applying insecticides and releasing sterile males to the environment where they mate with wild females but don't produce offspring. Ultimately, we'd like to contribute to the World Health Organization's goal of eliminating African sleeping sickness by 2030 with a new way to prevent the transmission of disease-causing trypanosomes to people and animals.![]()
- Author: Kathy Keatley Garvey

Bentley serves as the director of training and education for the National Pest Management Association (NPMA), headquartered in Fairfax, Va., and hosts NPMA's BugBytes. Kimsey, a global authority on wasps, bees and other insects, is a two-term past president of the International Society of Hymenopterists.
Kimsey fielded questions on the history of the hornet, its biology, its range, its behavior, its stings, and the news media frenzy over two reported incidents in North America. A single colony of the Asian giant hornet (AGH), Vespa mandarinia, was found and destroyed Sept. 18, 2019 in Nanaimo, Vancouver Island, Canada, and a single dead hornet was found Dec. 8, 2019 in nearby Blaine, Wash.
Concerned beekeepers worried that the hornets could become established and decimate their hives. Citizens throughout the country began reporting scores of "murder hornets," which turned out to be yellow jackets, European paper wasps, hover flies, hoverflies, moths and even a Jerusalem cricket (potato bug).
In the podcast, Kimsey relates that the Asian giant hornets are native to Asia, where the residents tolerate them. The beekeeping industry in Washington state, however, was "convinced that they are killing our honey bees," Kimsey told Bentley. "There's no basis in reality as far as I can tell," she said.
The Asian giant hornet is "one of about a dozen or so species in this genus," Kimsey said. She described them as "comically large and menacing looking."
The specimens in the Bohart Museum of Entomology are about 1.5 inches long. "I've never seen one two inches long. But it's a big animal--no question about it."
Bentley also discussed entomologist Justin Schmidt's Sting Pain Index, which rates the painful stings of some 83 hymenopteran species.
Kimsey agreed that the Asian giant hornet "can deliver a lot of venom" and "can sting repeatedly." But in her opinion, "the honey bee sting is the worst."
Other points Kimsey brought out included:
- The Asian giant hornets probably arrived here in cargo ships
- The larvae and pupae are restaurant-fare in some parts of Asia and are quite the delicacy
- The coronavirus pandemic has resulted in fewer cargo ships arriving in the United States from Asia, and thus fewer opportunities for hitchhikers.
Related Links (Bug Squad blog)
- About Those Asian Hornets (May 4, 2020)
- The Hornet Wars: 'A Bloody Dumpster Fire" (May 5, 2020)
- How Do You Say Murder Hornets? Delicious (May 8, 2020)
Matan Shelomi, former graduate student of Lynn Kimsey's and now an assistant professor of entomology at the National Taiwan University in Taipei, Taiwan, enjoys them. - Incredible Work, and Timely, on 22 Species of Hornets (May 12, 2020)

- Author: Kathy Keatley Garvey

Researchers from Harvard Medical School and the University of California, Davis, blocked the progression of cancer growth caused by environmental carcinogens and food contaminants by resolving an eicosanoid/cytokine storm triggered by cell debris.
The research, from the laboratories of physician-researcher Dipak Panigrahy of Harvard Medical School and UC Davis distinguished professor Bruce Hammock, is published in the current edition of the Proceedings of the National Academy of Sciences.
“We advanced the hypothesis that cell debris from chemotherapy, resection of tumors and even immunotherapy can make these therapies a double-edged sword stimulating cancer growth and metastasis while treating it,” said Hammock, who holds a joint appointment in the UC Davis Department of Entomology and Nematology and the UC Davis Comprehensive Cancer Center.
In their paper, “Resolution of Eicosanoid/Cytokine Storm Prevents Carcinogen and Inflammation-Initiated Hepatocellular Cancer Progression,” the scientists covered the potent environmental carcinogen and food contaminant aflatoxin. Aflatoxins are toxins produced by certain fungi that are found in such agricultural crops as corn, peanuts, cottonseed, and nuts.
“Not only is this fungal metabolite genotoxic but it is also a tumor promoter,” said Hammock, defining a genotoxic agent as “a chemical that damages cellular DNA, resulting in mutations or cancer.”

Lead authors Anna Fishbein of Harvard University, a recently enrolled medical student in the Georgetown University School of Medicine, and Weicang Wang, a postdoctoral scholar in the Hammock lab, said aflatoxin exerts some of its cancer-promoting effects by generating cell debris which activate a pathway leading to eicosanoid and cytokine storms. These two classes of natural chemical mediators, they explained, control many of our defenses against pathogens, but when out of control, these storms lead to growth and metastasis of liver cancer.
“We demonstrated that debris generated by aflatoxin B1accelerates tumor dormancy escape in liver cancer models by stimulating a macrophage-derived eicosanoid and cytokine storm of pro-inflammatory mediators,” said Fishbein. “Thus, targeting a single inflammatory mediator or eicosanoid pathway is unlikely to prevent carcinogen-induced tumor progression.”
The researchers showed that the inhibition of the soluble epoxide hydrolase (sEH) pathway or the combined inhibition sEH and cyclooxygenase-2 (COX-2) pathways prevented the carcinogen debris-induced storm of both cytokines and lipid mediators by macrophages--specialized detect-and-destroy cells.
In animal models, the dual COX-2/sEH inhibitor PTUPB prevented the onset of debris-stimulated liver cancer. The dual inhibition of COX-2/sEH pathways may be “a novel approach” to control cancer of the liver, the researchers said.

“We also showed that carcinogen-generated debris stimulates an endoplasmic reticulum (ER) stress response which may promote HCC progression. Importantly, PTUPB prevents the ER stress response,” Wang added. “We created a novel model of debris-stimulated liver cancer designed to study new strategies for the prevention and treatment of carcinogen-induced cancers with tremendous potential to translate to the clinic.”
From a nutritional standpoint, aflatoxin is a common food contaminant, Wang said. “But good agricultural practice and post-harvest technology keep the levels very low. However, in much of the world, aflatoxin levels are so high that many crops are discarded. In other cases, these contaminated grain and nut crops enter the human food chain, where they cause acute toxicity, severe anemia and of course later lead to cancer.”
UC Davis co-author and nutritional scientist Yuxin Wang (who is the wife of Weicang Wang) said that “finding a way to modulate the events that lead to the eicosanoid storm would have a major effect on children's health in many developing countries.”

Fishbein and Allison Gartung of the Panigrahy lab not only used the soluble epoxide hydrolase inhibitors from the Hammock lab but also used some prototype drugs synthesized by chemist Sung Hee Hwang of the UC Davis School of Veterinary Medicine “which proved to be even better,” Hammock said.
“These compounds are a synthetic combination of cyclooxygenase inhibitors like celebrex with epoxide hydrolase inhibitors,” Hammock said. “Since epoxide hydrolase inhibitors stabilize the endoplasmic reticulium stress response and transcriptionally down regulate inflammatory cyclooxygenase we expected them to synergize with cyclooxygenase inhibitors. We were surprised and pleased with the dramatic interaction of these inhibitors when combined in the same molecule in reducing the cytokine and eicosanoid production by in response to cell debris.”
“The observations from Harvard show that by inhibiting soluble epoxide hydrolase, we can block the activation of these inflammatory cascades leading to tumor promotion, growth and metastasis,” Hammock said. “We have a compound in human clinical trials that inhibits sEH, which should be clinically available in a few years. In addition. we have found natural inhibitors of the epoxide hydrolase in a variety of plants, including crop plants. This may allow us to reduce the cancer risk and block the gastrointestional erosion and bleeding caused by dietary aflatoxin using natural means.”
Other members of the 15-member team are UC Davis researchers Jun Yang, Yuxin Wang and Sung Hee Hwang; Harvard researchers Haixia Yang, Victoria Hallisey, Jianjun Deng, Sanne Verheul, Allison Gartung, Diane Bielenberg and Mark Kiernan (now of Bristol-Myers Squibb); and Sui Huang, Institute for Systems Biology, Seattle. Hammock and Panigrahy are the corresponding authors.
The research drew strong financial support as the Panigrahy's laboratory is generously supported by the Credit Unions Kids at Heart Team, the CJ Buckley Pediatric Brain Tumor Fund, and the Joe Andruzzi Foundation; and Hammock's UC Davis grants from the National Institute of Environmental Health (NIEHS) Superfund Research Program, and the NIEHS RIVER Award (Revolutionizing Innovative, Visionary, Environmental Health Research).
Hammock, a member of the UC Davis faculty since 1980, has directed the UC Davis Superfund Research Program for nearly four decades. It supports scores of pre- and postdoctoral scholars in interdisciplinary research in five different colleges and graduate groups on campus. Last year Hammock received a $6 million, eight-year “Outstanding Investigator” federal grant for his innovative and visionary environmental health research: The award is part of the Revolutionizing Innovative, Visionary Environmental Health Research (RIVER) Program of NIEHS.
The abstract:
“Toxic environmental carcinogens promote cancer via genotoxic and nongenotoxic pathways, but nongenetic mechanisms remain poorly characterized. Carcinogen-induced apoptosis may trigger escape from dormancy of microtumors by interfering with inflammation resolution and triggering an endoplasmic reticulum (ER) stress response. While eicosanoid and cytokine storms are well-characterized in infection and inflammation, they are poorly characterized in cancer. Here, we demonstrate that carcinogens, such as aflatoxin B1 (AFB1), induce apoptotic cell death and the resulting cell debris stimulates hepatocellular carcinoma (HCC) tumor growth via an ‘eicosanoid and cytokine storm.' AFB1-generated debris up-regulates cyclooxygenase-2 (COX-2), soluble epoxide hydrolase (sEH), ER stress-response genes including BiP, CHOP, and PDI in macrophages. Thus, selective cytokine or eicosanoid blockade is unlikely to prevent carcinogen-induced cancer progression. Pharmacological abrogation of both the COX-2 and sEH pathways by PTUPB prevented the debris-stimulated eicosanoid and cyto- kine storm, down-regulated ER stress genes, and promoted macrophage phagocytosis of debris, resulting in suppression of HCC tumor growth. Thus, inflammation resolution via dual COX-2/sEH inhibition is an approach to prevent carcinogen-induced cancer.”


