- Author: Alejandro Del Pozo-Valdivia
We are happy to announce that the diamondback moth capture data, presented as maps, is now housed in our own University of California Cooperative Extension Monterey website.
To access to these maps, simply click on the link below:
http://cemonterey.ucanr.edu/Agriculture/2019_Diamondback_moth_monitoring_maps/
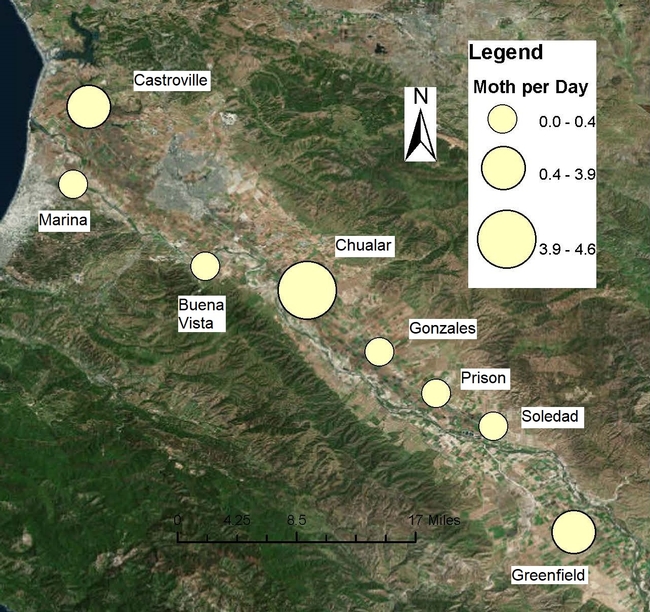
These maps use the closest town or landmark where the traps are located to show moths per trap per day. Moth captures are presented as yellow bubbles. The bigger the bubble, the larger the population of moths is.
On the same page, you will also find the overall population fluctuations of these moths in the Valley, as a series chart.
We also stored the overall fluctuation of aphids and thrips numbers, captured in yellow sticky cards in our UCCE Monterey website. To access to these bar charts, click on the link below:
http://cemonterey.ucanr.edu/Agriculture/2019_Aphid_and_Thrips_Monitoring_Program/
If you would like to learn more about these three monitoring programs happening in the Salinas Valley, do not hesitate to contact Alejandro Del-Pozo at adelpozo@ucanr.edu or 831-759-7359.
- Author: Michael D Cahn
- Author: David Chambers
A tensiometer is a very useful tool for monitoring soil moisture status of vegetable and berry crops. Compared to other sensors that often require equipment such as dataloggers or a computer to collect readings, tensiometers can be easily read by irrigators in the field. Also, tensiometer readings are not affected by variations in soil texture, temperature, and salinity and they can operate without electricity (no batteries needed).
What is tension? Tensiometers measure soil moisture in units of negative pressure also known as tension. Tension is a measure of the force that plant roots need to exert to pull water from the soil pores. Large pores hold water with less force than small pores. As plants extract moisture from the soil, water is first taken up from the largest pores. As the soil dries roots need to exert more force to pull water from the smaller pores. Hence, high tension values mean that the soil is becoming dry.
How do tensiometers work? Tensiometers are filled with water (preferably distilled) that has been degassed by boiling. A key component of the tensiometer is a porous ceramic cup which allows water in the shaft of the tensiometer to freely pass into the soil without air bleeding though the small pores in the cup (Fig. 1). If the soil is not saturated, water will move from inside the cup into the unfilled soil pores. Because air cannot replace the space vacated by the exiting water, a vacuum develops in the shaft of the tensiometer that can be measured with an accurate gauge. Water will stop migrating from inside the tensiometer cup into the soil when the internal vacuum pressure of the tensiometer equals the soil tension, or the force needed to pull water from the soil pores. The vacuum gauge measures tension in units of kPa or cbars, which are equivalent (1 kPa = 1 cbar).
Interpretation of tension readings Because the tension value provides a sense of how much energy a plant would need to exert to suck water from the soil, tensiometer readings can be easily related to water stress in crops. At high tension values a plant experiences more water stress and growth slows. In addition, a tension reading has a similar meaning in terms of water stress whether the soil has a sandy, clay or loam texture.
Reliability of tensiometers The one Achilles' heal or weakness of the tensiometer is that if any air leaks into the instrument it will not retain a vacuum and the readings will be unreliable. There are several brands of commercial tensiometers available. Some are relatively inexpensive and simple to use, and others are more complex and can be interfaced with dataloggers to provide continuous readings throughout the day. Based on our experience, some of the most popular commercially available tensiometers often leak air and lose vacuum pressure, and in many cases the gauges do not provide accurate readings or are not durable. The loss of vacuum pressure means that the tensiometers need to be frequently refilled with degassed water. Also, irrigators may mistake a low reading to indicate that a crop has adequate moisture when in reality the soil may be dry.
A dependable tensiometer design We designed and tested a version of a tensiometer in 2018 that was simple to build and provided accurate readings for a material cost of less than $55 . The design improved the ability of the instrument to retain a vacuum at high tensions. Under moderately moist soil conditions the tensiometer usually required refilling with degassed water less than once per month. Even when the soil dried to tensions above the maximum range of the tensiometer (> 80 kPa), these tensiometers continued to hold a vacuum for about two weeks until all of the water in the shaft was depleted.
The following paragraphs describe the materials needed (Fig. 2) and procedures to build a tensiometer. The vendors of the materials are examples of ones that we use, but you may identify different or cheaper sources for these components. By carefully following these instructions, one should be able to build a dependable tensiometer that provides accurate tension readings. An update to this design can also be found in a more recent blog article.
Materials needed:
Ceramic cups
Vender: SoilMoisture Equipment Corporation, Santa Barbara CA (805-964-3525) Part Number 0655X01-B01M3, Dimensions: 0.875 inch OD x 2.75 inch length. Cost: $30.80 ea.
Epoxy (ceramic/plastic)
Vender: SoilMoisture Equipment Corporation, Santa Barbara CA (805-964-3525)
Part Number 0980V004, Description: 4 oz: epoxy and 4 oz hardener. Cost: $106 ea. Note that the epoxy/hardener is a sufficient volume to make several hundred tensiometers.
Vacuum gauge
Vender: Zoro.com/Grainger.com Part Number 4FMK3, Description: ¼ inch MNPT 2 inch diameter test vacuum gauge. Cost: $18.09 ea.
#1 size rubber stopper
Vender: Grainger.com Part Number 8DWU6, model RST1-S, Description: 24 mm neck, bottom diam. = 14 mm. Top diam. = 20 mm. Cost: $18.08 / 52 pieces
Schedule 40 PVC pipe (½ inch diameter) Vender: irrigation supply or hardware store
PVC “T”
Vender: irrigation supply or hardware store, Manufacturer: Spears Inc. Part number 402-072, Description: ½ inch slip x ¼ inch threaded reducing "T."
PVC glue (gray) and purple primer
Vender: irrigation supply or hardware store
Gas pipe thread sealant (white or blue paste type)
Vender: irrigation supply or hardware store
Painters masking tape
Vender: hardware store
Petroleum Jelly (Vasoline)
Vender: pharmacy
Tools needed:
- PVC saw or PVC cutting tool
- Aluminum Oxide grinding stone, Manufacturer: Forney Part Number: A11 60028 Description: 7/8 in [23 mm] diam. x 2 inch [50.8mm] length
- Power hand-held drill
- Miter box
- Pocket knife
Procedures
1. Cut PVC pipe sections in the following lengths
1 foot depth tensiometer: top shaft = 4 inches, bottom shaft = 17 inches
2 foot depth tensiometer: top shaft = 4inches, bottom shaft = 30 inches
It is advisable to cut the bottom shaft about 1-inch longer than indicated above and then carefully cut the lower end of the shaft using the miter box or electric miter saw to assure that it is cut at a 90-degree angle. The ceramic cup will fit crooked on the end of the shaft if the cut deviates from 90 degrees.
- First glue the top shaft and then the bottom shaft to the ½ PVC “T” using the PVC glue. Make sure that you do not glue the end of the bottom shaft that was trimmed to 90 degrees. In a well-ventilated location, apply PVC primer to both the end of the shaft and the inside of the “slip” end of the “T”. Then apply gray PVC glue to both sides, and push the parts together, and hold in place for about 30 seconds to 1 minute. Tip: slightly twist the parts by about 30 degrees immediately after gluing to assure that the parts are secure. Also cover the non-glued areas with painter's tape to prevent the outside from becoming covered with glue.
- Slightly bevel the inside of the lower end of the bottom shaft using the handheld drill and grinding stone (Fig. 3). Alternatively, one can use a knife to bevel the end. Whether using the drill or the knife to bevel the inside of the pipe, stop periodically and test fit the ceramic cup. This way you will not remove too much material, and will quickly get a feel for the appropriate amount to remove.
 Figure 3. A drill is used to bevel the lower end of the tensiometer shaft to accommodate the ceramic cup.
Figure 3. A drill is used to bevel the lower end of the tensiometer shaft to accommodate the ceramic cup. - Use epoxy to glue the ceramic cup to the lower end of the bottom shaft. Protect the ceramic cup during the gluing process by covering the outside with painter's tape (Fig. 4). Check that the ceramic cup fits snuggly into the PVC tube and is aligned straight. If using the epoxy from SoilMoisture equipment epoxy, mix up 1-part epoxy with 1-part hardener. Mix thoroughly. Only a small amount of epoxy is needed to coat the throat of the ceramic cup and the inside of the PVC tube, so it may be best to glue several tensiometers at the same time so that the epoxy is not wasted. One can usually glue no more than 20 to 40 cups at a time becaue the epoxy begins to cure after an hour. Approximately 20 ml of epoxy is needed for 20 tensiometers. The cure time is temperature dependent. Full cure is 8 hours at 77 °F. It is best to allow more time for curing. After gluing, painter's tape can be used to secure the cup to the shaft. Take care when securing the two with the tape to assure that the cup is aligned with the PVC shaft. Let the glue set for at least 24 hours with the tensiometer supported with the cup-end up in a vertical position. Tip: best if parts are glued at temperatures above 65 °F. More hardener may be needed at lower temperatures. Also, it is advisable to first test a small batch of epoxy to assure that the proportion of hardener to epoxy is enough for epoxy to set up hard.
 Figure 4. Use painter’s tape to prevent glue from coating the PVC shaft and ceramic cup.
Figure 4. Use painter’s tape to prevent glue from coating the PVC shaft and ceramic cup. - Coat the ¼ inch male threads of the gauge with pipe thread sealant and hand screw on the vacuum gauge. Tip: do not over tighten or the PVC “T” will crack!
- Fill the tensiometer fully with degassed distilled water. The water can be degassed by boiling it and allowing it to cool.
- Coat the lower end of the rubber stopper with a thin film of petroleum jelly and insert into the top end of the tensiometer with a light twist to firmly seat the stopper (A loose stopper is the main cause for vacuum leaks).
 Figure 5. Finished tensiometer is ready for testing.
Figure 5. Finished tensiometer is ready for testing.
Preparing the tensiometer for testing and field installation
The tensiometer should be filled with degassed water (preferably distilled) before testing. Tap water will work too, but if it is hard water (contains a high concentrations of calcium and carbonate) it could cause precipitates to form inside the ceramic cup. Boiling will expel much of the dissolved air from the water. We do not recommend using a vacuum pump to remove dissolved air from the water. Boiling works best for degassing water. It also helps to soak the ceramic cup end of the tensiometer in water for a few hours so that the pores of the ceramic cup are saturated before testing and/or installation.
Testing the tensiometer for air (vacuum) leaks
After filling the tensiometer with water and sealing it with a rubber stopper, wrap a dry paper towel on the end of the ceramic cup and hold it tightly (Fig. 6). If the tensiometer is filled with degassed water, the tension should quickly increase to about 20 to 30 kPa as the towel absorbs water from the cup. If the gauge does not increase above 0, air is likely leaking into the tensiometer. Check the glue joints and assure that the stopper is tightly in place.
If the tension quickly increases to more than 20 kPa, then leave the tensiometer out in the sun to assure that the tension rises to above 70 to 80 kPa. This may take some time, minutes to hours, depending on the ambient temperature. If the tension does not increase to a high value, then check glue joints and the stopper. Also check that the gauge is securely threaded into the PVC “T.”

Installing tensiometers in the field
Proper installation of a tensiometer in the field will achieve close contact between the ceramic cup and surrounding soil. Using a soil probe with a ½ inch diameter shaft, make a pilot hole to a depth a few inches shallower that the depth of installation (Fig. 7). Make a soil water slurry by thoroughly mixing soil with the water to a pancake batter-like consistency. Add some slurry into the hole and push the tensiometer to the desired depth (Fig. 8). The soil slurry assures that water can freely move between the ceramic cup and the surrounding soil and fills the voids between the hole and tensiometer shaft. Formation of air gaps between the ceramic cup and the soil will lessen the accuracy of tensiometer readings. After two days of equilibration, the tensiometer reading should accurately reflect the tension of the soil.

- Author: Alejandro Del Pozo-Valdivia
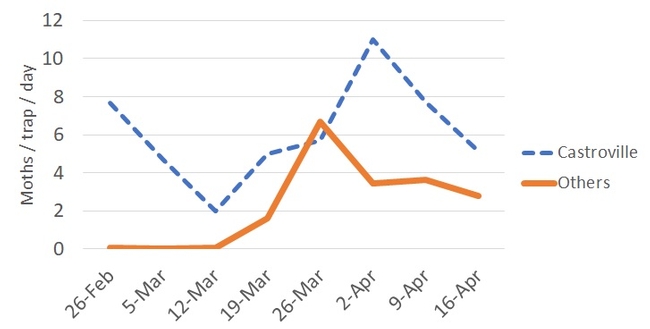
We continue the effort of monitoring diamondback moth (DBM) across the Salinas Valley using sex pheromone baited traps, as shown in the picture above. We have been adding additional traps to cover a larger area along Highway 101. We have daily moth capture data from Castroville to Greenfield. The chart below summarizes these daily captures from our pheromone traps.
As stated in a previous blog post, higher numbers of DBM have been usually recorded in Castroville. It seems like we just passed a generation of adults during late March to early April. Currently, DBM capture numbers are going down. Lower adults in the system could be paired with more caterpillars feeding on several host plants.
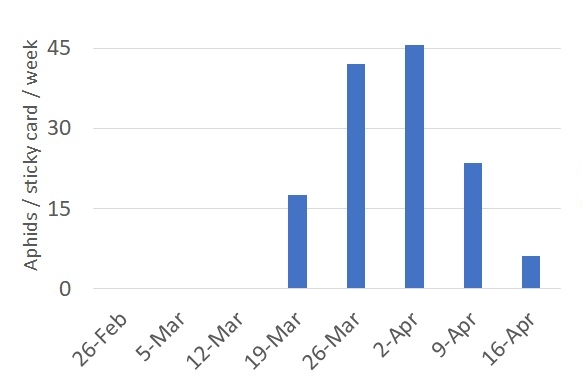
We have set up yellow sticky cards to track the overall population of winged aphids and thrips. These sticky cards are in the same locations as the pheromone traps. At this point, data for aphids and thrips is not broken down at the species level.
From the chart above, there was a flight of aphids during late March to early April. Some PCAs mentioned to me that foxglove aphid started to show up in their fields during that time period. It seems now that winged aphid numbers are going down. However, it does not mean that numbers of aphids in our crops are decreasing. We might be facing higher population of wingless aphids in our crops right now.
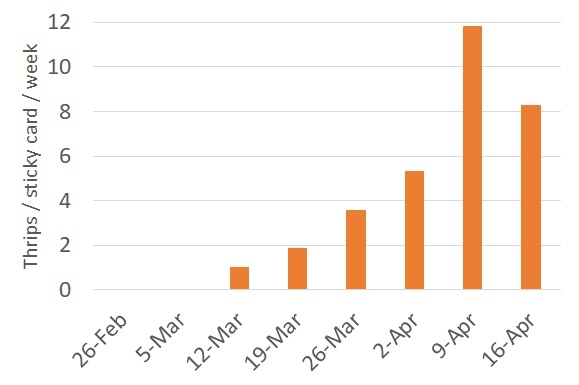
We need to keep an eye on population dynamics of thrips in the Valley. The ultimate goal is to be better prepared this season to manage those creatures and reduce the incidence of INSV virus. The chart below shows captures of thrips in our sticky cards.
It seems like thrips populations had a spike two weeks ago. Currently, thrips numbers are going down. I believe that keeping track of the fluctuation of thrips numbers in our Valley would help us detect large populations of this pest. There is a need to alert PCAs when the front of a 'thrips wave' would happen.
If you are interested in getting more information on this monitoring effort, please do not hesitate in contacting Alejandro Del-Pozo at 831-759-7359 or adelpozo@ucanr.edu.
- Author: Surendra K. Dara
Integrated pest management, commonly referred to as IPM, is a concept of managing pests that has been in use for several decades. The definition and interpretation of IPM vary depending on the source, such as a university, institute, or a researcher, and its application varies even more widely depending on the practitioner. Here are a few examples of its definitions and interpretations:
“IPM is an ecosystem-based strategy that focuses on long-term prevention of pests or their damage through a combination of techniques such as biological control, habitat manipulation, modification of cultural practices, and use of resistant varieties. Pesticides are used only after monitoring indicates they are needed according to established guidelines, and treatments are made with the goal of removing only the target organism. Pest control materials are selected and applied in a manner that minimizes risks to human health, beneficial and nontarget organisms, and the environment.” UC IPM
“Integrated Pest Management, or IPM, is an approach to solving pest problems by applying our knowledge about pests to prevent them from damaging crops, harming animals, infesting buildings or otherwise interfering with our livelihood or enjoyment of life. IPM means responding to pest problems with the most effective, least-risk option.” IPM Institute of North America
“A well-defined Integrated Pest Management (IPM) is a program that should be based on prevention, monitoring, and control which offers the opportunity to eliminate or drastically reduce the use of pesticides, and to minimize the toxicity of and exposure to any products which are used. IPM does this by utilizing a variety of methods and techniques, including cultural, biological and structural strategies to control a multitude of pest problems.” Beyond Pesticides
“IPM is rotating chemicals from different mode of action groups.” A grower
These definitions and interpretations represent a variety of objectives and strategies for managing pests. IPM is not a principle that can/should be strictly and equally applied to every situation, but a philosophy that can guide the practitioner to use it as appropriate for the situation. For example, varieties that are resistant to arthropod pests and diseases are available for some crops, but not for others. Mating disruption with pheromones is widely practiced for certain lepidopteran and coleopteran pests, but not for several hemipteran pests. Biological control is more readily employed for greenhouse pests, but not to the same extent under field conditions. While chemical pesticides should be used as the last resort, in principle, sometimes they are the first line of defense to prevent damage to the transplants by certain pests or area-wide spread of certain endemic or invasive pests and diseases.
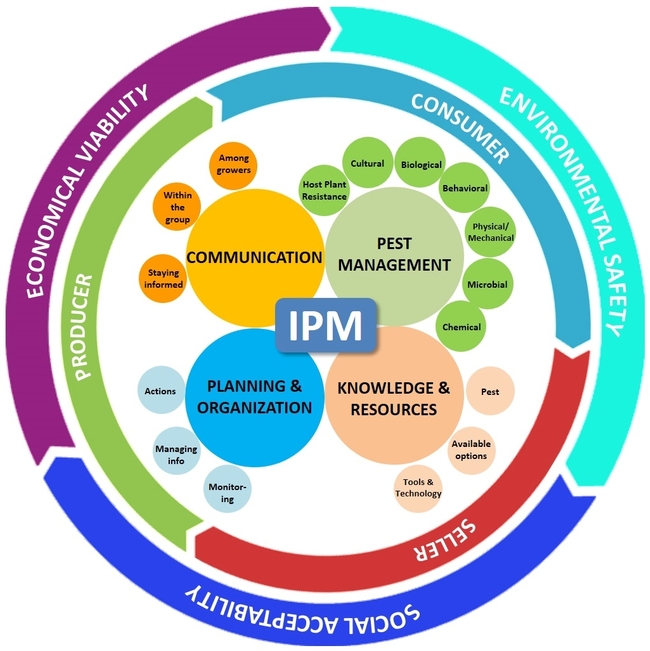
Crop production is an art, science, and business, and by adding environmental and social factors, IPM – an approach used in agriculture – can also be influenced by a number of factors. Each grower has their own strategy for producing crops, minimizing losses, and making a profit in a manner that is acceptable to the society, safe for the consumers, and less disruptive to the environment. In other words, “IPM is an approach to manage pests in an economically viable, socially acceptable, and environmentally safe manner”. Keeping this simple, but loaded, definition in mind and considering recent advances in crop production and protection, communication technology, and globalization of agriculture and commerce, here is the new paradigm of IPM with its management, business, and sustainability aspects.
I. Management Aspect
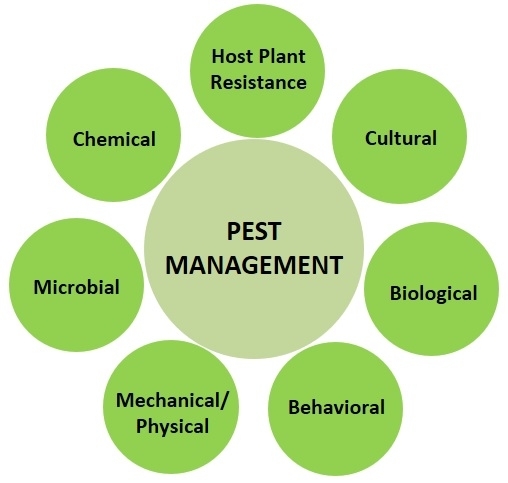
There are four major components in the IPM model that address the various pest management options, the knowledge and resources the grower has in order to address the pest issue, planning and organization of information to take appropriate actions, and maintaining good communication to acquire and disseminate knowledge about pests and their management.
1. Pest Management:
The concept of pest control has changed to pest management over the years knowing that a balanced approach to managing pest populations to levels that do not cause economic losses is better than eliminating for environmental and economic reasons. Although the term control is frequently used in literature and conversations, it generally refers to management. A thorough knowledge of general IPM principles and various management options for all possible pest problems is important as some are preventive and others are curative. It is also essential to understand inherent and potential interactions among these management options to achieve maximum control. The following are common control options that can be employed at different stages of crop production to prevent, reduce, or treat pest infestations. Each of them may provide only a certain level of control, but their additive effect can be significant in preventing yield losses.
a. Host plant resistance: It involves the use of pest resistant and tolerant cultivars developed through traditional breeding or genetic engineering. These cultivars possess physical, morphological, or biochemical characters that reduce the plant's attractiveness or suitability for the pest to feed, develop, or reproduce successfully. These cultivars resist or tolerate pest damage and thus reduce the yield losses.
b. Cultural control: Changing agronomic practices to avoid or reduce pest infestations and damage refers to cultural control. Adjusting planting dates can help escape pest occurrence or avoid most vulnerable stages. Modifying irrigation practices, fertilizer program, plant or row spacing, and other agronomic practices can create conditions that are less suitable for the pest. Destroying crop residue and thorough cultivation will eliminate breeding sites and control soil-inhabiting stages of the pest. Crop rotation with non-host or tolerant crops will break the pest cycles and reduce their buildup year after year. Choosing clean seed and plant material will avoid the chances of introducing pests right from the beginning of the crop production. Sanitation practices to remove infected/infested plant material, regular cleaning field equipment, avoiding accidental contamination of healthy fields through human activity are also important to prevent the pest spread. Intercropping of non-host plants or those that deter pests or using trap crops to divert pests away from the main crop are some of the other cultural control strategies.
c. Biological control: Natural enemies such as spiders, predators, and parasitic wasps can be very effective in causing significant reductions in pest populations in certain circumstances. Periodical releases of commercially available natural enemies or conserving natural enemy populations by providing refuges or avoiding practices that harm them are some of the common practices to control endemic pests. To address invasive pest issues, classical biological control approach is typically employed where natural enemies from the native region of the invasive pest are imported, multiplied, and released in the new habitat of the pest. The release of irradiated, sterile insects is another biological control technique that is successfully used against a number of pests.
d. Behavioral control: Behavior of the pest can be exploited for its control through baits, traps, and mating disruption techniques. Baits containing poisonous material will attract and kill the pests when distributed in the field or placed in traps. Pests are attracted to certain colors, lights, odors of attractants or pheromones. Devices that use one or more of these can be used to attract, trap or kill pests. Pheromone lures confuse adult insects and disrupt their mating potential, and thus reduce their offspring.
e. Physical or mechanical control: This approach refers to the use of a variety of physical or mechanical techniques for pest exclusion, trapping (in some cases similar to the behavioral control), removal, or destruction. Pest exclusion with netting, handpicking or vacuuming to remove pests, mechanical tools for weed control, traps for rodent pests, modifying environmental conditions such as heat or humidity in greenhouses, steam sterilization or solarization, visual or physical bird deterrents such as reflective material or sonic devices are some examples for physical or mechanical control.
f. Microbial control: Using entomopathogenic bacteria, fungi, microsporidia, nematodes, and viruses, and fermentation byproducts of microbes against arthropod pests, fungi against plant parasitic nematodes, and bacterial and fungal antagonizers of plant pathogens generally come under microbial control.
g. Chemical control: Chemical control typically refers to the use of synthetic chemical pesticides, but to be technically accurate, it should include synthetic chemicals as well as chemicals of microbial or botanical origin. Although botanical extracts such as azadirachtin and pyrethrins, and microbe-derived toxic metabolites such as avermectin and spinosad are regarded as biologicals, they are still chemical molecules, similar to synthetic chemicals, and possess many of the human and environmental safety risks as chemical pesticides. Chemical pesticides are categorized into different groups based on their mode of action and rotating chemicals from different groups is recommended to reduce the risk of resistance development. Government regulations restrict the time and amount of certain chemical pesticides and help mitigate the associated risks.
The new RNAi (ribonucleic acid interference) technology where double-stranded RNA is applied to silence specific genes in the target insect is considered as a biopesticide application. Certain biostimulants based on minerals, microbes, plant extracts, seaweed or algae impart induced systemic resistance to pests and diseases, but are applied as amendments without any claims for pest or disease control. These new products or technologies can fall into one or more abovementioned categories.
As required by the crop and pest situation, one or more of these control options can be used throughout the production period for effective pest management. When used effectively, non-chemical control options delay, reduce, or eliminate the use of chemical pesticides.
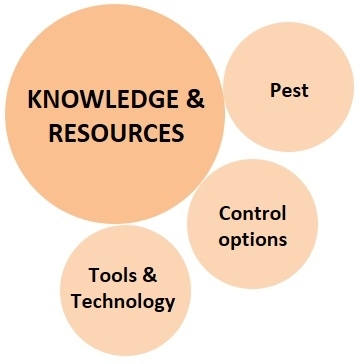
2. Knowledge and Resources:
The knowledge of various control options, pest biology and damage potential, and suitability of available resources enables the grower to make a decision appropriate for their situation.
a. Pest: Identification of the pest, understanding its biology and seasonal population trends, damaging life stages and their habitats, nature of damage and its economic significance, vulnerability of each life for one or more control options, host preference and alternate hosts, and all the related information is critical for identifying an effective control strategy.
b. Available control options: Since not all control options can be used against every pest, the grower has to choose the ones that are ideal for the situation. For example, systemic insecticides are more effective against pests that mine or bore into the plant tissue. Pests that follow a particular seasonal pattern can be controlled by adjusting planting dates. Commercially available natural enemies can be released against some, while mating disruption works well against others. Entomopathogenic nematodes can be used against certain soil pests, bacteria and viruses against pests with chewing mouthparts such as lepidoptera and coleopteran, and fungi against sucking pests.
c. Tools and technology: A particular pest can be controlled by certain options, but they may not all be available in a particular place, for a particular crop, or within the available financial means. For example, the release of natural enemies may be possible in high-value speciality crops, but not in large acreage field crops. A particular pesticide might be registered against a pest on some crops, but not on all. Use of netting or tractor-mounted vacuums can be effective, but very expensive limiting their availability to those who can afford.
This is a critical component where diagnostic and preventive or curative decisions are made based on available and affordable control options.
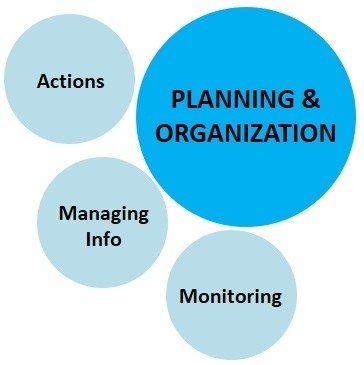
3. Planning and Organization:
This component deals with the management aspect of the of the new IPM model for data collection, organization, and actual actions against pest infestations.
a. Pest monitoring: Regularly monitoring the fields for pest infestation and spread is a basic step in crop protection. Early detection in many cases can help address the pest situation by low-cost spot treatment or removal of pests or infected/infested plant material. When pest infestations continue to grow, regular monitoring is necessary to assess the damage and determine the time to initiate farm-wide control. Monitoring is also important to avoid calendar-based pesticide applications especially at lower pest populations that do not warrant treatments.
b. Managing information: A good recordkeeping about pests, their damage, effective treatments, seasonal fluctuations, interactions with environmental factors, irrigation practices, plant nutrition, and all related information from year to year will build the institutional knowledge and prepares the grower to take preventive or curative actions.
c. Corrective actions: Taking timely action is probably the most important aspect of IPM. Even with all the knowledge about the pest and availability of resources for its effective management, losses can be prevented only when corrective actions are taken at the right time. Good farm management will allow the grower to take timely actions. These actions are not only necessary to prevent damage on a particular farm, but also to prevent the spread to neighboring farms. When pest management is neglected, it leads to area-wide problems with larger regulatory, social, and economic implications.
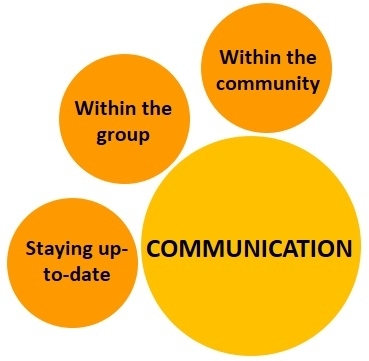
4. Communication:
Good communication to transfer the individual or collective knowledge for the benefit of everyone is the last component of the new IPM model. Modern and traditional communication tools can be used for outreach as university and private researchers develop information about endemic and invasive pests, emerging threats, and new control strategies.
a. Staying informed: Growers and pest control professionals should stay informed about existing and emerging pests and their management options. Science-based information can be obtained by attending extension meetings, webinars, or workshops, reading newsletter, trade, extension, or scientific journal articles, and keeping in touch with researchers and other professionals through various communication channels. Well-informed growers can be well prepared to address pest issues.
b. Communication within the group: Educating farm crew through periodical training or communication will help with all aspects of pest management, proper pesticide handling, ensuring worker safety, and preventing environmental contamination. Knowledgeable field crew will be beneficial for effective implementation of pest management strategies.
c. Communication among growers: Although certain crop production and protection strategies are considered proprietary information, pests do not have boundaries and can spread to multiple fields when they are not effectively managed throughout the region. Sharing knowledge and resources with each other will improve pest control efficacy and benefit the entire grower community.
In addition to these four components with an IPM model, factors that influence profitable, safe, and affordable food production at a larger scale and their implications for global food security should also be included. There are two layers surrounding these four components addressing the business and sustainable aspects of food production.
II. Business Aspect:
Consumers want nutritious, healthy, and tasty produce that is free of pest damage at affordable prices. Growers try to meet this demand by producing food that meets all the consumer needs, while maintaining environmental and human safety and still being able to make a profit. Sellers evaluate the market demand and strategize their sales to satisfy consumers while making their own profit to stay in the business. In an ideal system, consumer, producer, and seller would maintain a harmonious balance of food production and sale. In such a system, food is safe and affordable to everyone, there will be food security all over the world, and both growers and sellers make a good profit with no or minimal risk to the environment in the process of food production. However, this balance is frequently disrupted due to i) consumers' misunderstanding of various food production systems, their demand for perfectly shaped fruits and vegetables at affordable prices or their willingness to pay a premium price for food items that are perceived to be safe, ii) growers trying to find economical ways of producing high quality food while facing with continuous pest problems and other challenges, and iii) sellers trying to market organic food at a higher price as a safer alternative to conventionally produced food. If growers implement good IPM strategies to produce safe food and consumers are aware of this practice and gain confidence in food produced in an IPM system, then sellers would be able to market what informed-consumers demand.
III. Sustainability Aspect:
As mentioned earlier, IPM is an approach to ensure economic viability at both consumer and producer level (seller is always expected to make a profit), environmental safety through a balanced use of all available pest control options, and social acceptability as food is safe and affordable.
While organic food production is generally perceived as safe and sustainable, the following examples can explain why it is not necessarily true. Organic food production is not pesticide-free and some of the pesticides used in an organic system are as harmful to humans and non-target organisms as some chemical pesticides. Certain organically accepted pesticides have toxins or natural chemical molecules that are very similar to those in synthetic pesticides. In fact, some synthetic pesticides are manufactured imitating the pesticidal molecules of natural origin. Mechanical pest control practices such as vacuuming or tilling utilize fossil fuels and indirectly have a negative impact on the environment. For example, diesel-powered tractors are operated for vacuuming western tarnished bug in strawberry 2-3 times or more each week while a pesticide application typically requires the use of tractor once every 7-14 days. To control certain pests, multiple applications of organic pesticides might be necessary with associated costs and risks, while similar pest populations could be controlled by fewer chemical pesticide applications. It is very difficult to manage certain plant diseases and arthropod pests through non-chemical means and inadequate control not only leads to crop losses, but can result in their spread to larger areas making their control even more difficult. Many growers prefer a good IPM-based production to an organic production for the ease of operation and profitability. However, they continue to produce organic food to stay in business.
While middle and upper-class consumers may be willing to pay higher prices for organically produced food, many of the low-income groups in developed and underdeveloped countries cannot afford such food. Organic food production can lead to social inequality and a false sense of wellbeing for those can afford. Food security for the growing world population is necessary through optimizing input costs, minimizing wastage, grower adoption of safe and sustainable practices, and consumer confidence in food produced through such practices. IPM addresses all the economic, environmental, and social aspects and provides safe and affordable food to the consumers and profits to producers and sellers, while maintaining environmental health.
- Author: Richard Smith
Leaf spots are a common issue that affects spinach and reduces its salability. Spots on spinach leaves are caused by both biotic and abiotic influences. Insects, such as leafminers, frequently cause spots on spinach leaves. Female leafminers stipple spinach leaves by puncturing the leaf surface with their ovipositors and then feeding on plant sap that exudes from the holes. The stippled areas often occur in clusters and have a characteristic look due to the broken epidermal cells in the center of the stipple (photo below).
Diseases that cause spots on lettuce include the following: Cladosporium causes round, tan lesions that have dark green spores and mycelia in the center of the spots (http://ipm.ucanr.edu/PMG/r732100311.html); Anthracnose lesions start as dark green water-soaked lesions that later turn tan with black fruiting bodies in the center (can be observed with a good hand lens, http://ipm.ucanr.edu/PMG/r732100211.html); Stemphylium causes circular lesions, but no fungal fruiting bodies or mycelia occur in the lesions making it difficult to distinguish this disease from abiotic causes (http://ipm.ucanr.edu/PMG/r732100411.html).

Abiotic leaf spots on spinach are caused by a variety of factors. The most common cause is from burn caused by herbicides, other pesticides or fertilizers; in addition, water and other stresses can cause spotting or lesions on spinach. Spinach leaves are quite sensitive to chemicals and will readily respond to them by developing chlorotic or tan colored necrotic areas. The size and distribution of the lesions can often provide clues as to the cause of the issue. For instance, spots caused by herbicides or other chemicals (Photos No. 1) often have a characteristic pattern which can reveal if the burn was caused by spray drift, herbicide on dust or even a direct spray. Spray drift or lift off of oxyfluorfen often causes small diffuse spotting on the leaves. The location of the affected plants in the field can indicate the direction that the chemical came from. Also, examining the pattern of the lesions on the plant can give an indication when the drift incident may have occurred, depending on which age of leaves are affected; for instance, younger leaves may be unaffected because they were protected down in the crown of the plant when the incident occurred. Also, if only the outer edge of leaves are affected, this may indicate that the base of the leaves were protected by shingling of other leaves; this symptom often helps to confirm that you are dealing with a drift issue vs some other cause. In many of these types of situations, having some background information about recent spray applications in the vicinity of the field helps piece together how and when the incident occurred. Spotting on the weeds also provides confirmation of the cause of the incident. Distortion of the leaves (Photos No. 2) occurs when the necrosis occurs early in the development cycle of the leaf; in this situation, the expanding young leaf continues to develop around the dead lesion and results in distorted growth. Chemical issues can also cause a sub-lethal response in spinach leaves which results in chlorotic lesions (Photos No. 3).
Other chemicals that commonly cause burn on spinach include salts. Salts in water can cause spotting and marginal burns on spinach (Photos No. 4), but it is rare for water to be salty enough to cause this issue. However, water-run injections of fertilizer are salty enough if the injected fertilizer is not adequately rinsed from the irrigation lines after the injection. Fertilizer burn from water runs typically look different than spray burn because the lesions often occur along the edge of the leaves and are typically larger and blotchier (Photos No. 5). Sometimes growers are surprised that their spinach was burned from fertilizer because they routinely apply fertilizer through the sprinkler with no issues; however, if the clean-out phase of the injection was not long enough, then fertilizer burn can readily occur. Topdress applications with dry fertilizer at the 1st – 2nd true leaf stage can also cause burn on spinach leaves if the fertilizer prills stick to moisture on the leaves (Photos No. 6).
Most of the spotting that we see on spinach is from the above-mentioned causes. However, other issues that can cause defects on the leaves include water stress (Photos No. 7) which is often tied to hot spells. Water stress characteristically will occur in large blotches in the interveinal region of the leaf. It mostly occurs on older to mid-aged leaves and on various locations on the leaf. Hail damage (Photo No. 8) occasionally occurs in the spring and causes light green spots due to damaged epidermal cells that lets the green from tissue lower in the leaf to show through. Tipburn of spinach occurs in the spring when air temperatures increase, but soil temperatures are still cool. Under such conditions a localized deficiency of calcium develops out on the tip of the young leaf; as the leaf continues to expand, the necrotic tissue on the tip inhibits expansion resulting in a “hooded” shape to the leaf (Photos No. 9). Occasionally, chimeras are seen in spinach fields which are the result of a mutation that occurs in the meristem of the leaf; the resulting leaves have a dramatic calico yellow and green appearance (Photos No. 9).
1. Herbicide or other Chemical Burn




2. Necrosis and Distortion
3. Yellow Lesions


4. Salts in Water
5. Water-run Fertilizer Burn
6. Dry, Topdress Fertilizer Burn
7. Water Stress

8. Hail Damage

9. Other Leaf Issues